|
The global 3D Printing in Healthcare Market is estimated to be valued at US$ 1.87 Bn in 2022 and is expected to exhibit a CAGR of 19.0% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights.
Market Overview: 3D printing in the healthcare industry refers to the usage of additive manufacturing technology to create three-dimensional objects, such as prosthetics, implants, medical devices, and pharmaceuticals. The advantages of 3D printing in healthcare include precision, customization, cost-effectiveness, and improved patient outcomes. The need for 3D printed products in the healthcare sector is driven by the rising demand for personalized medical treatments, growing geriatric population, and advancements in 3D printing technology. Market key trends: One key trend in the 3D Printing in Healthcare Market is the increasing adoption of 3D printing for the production of customized implants and prosthetics. With 3D printing, medical professionals can create personalized implants and prosthetics that are tailored to the individual patient's unique anatomy. This eliminates the need for off-the-shelf solutions and allows for better fitting, comfort, and functionality. Additionally, 3D printing enables the production of complex and intricate designs that would be difficult or impossible to achieve using traditional manufacturing methods. This trend is driven by the growing demand for patient-specific treatments and the advancements in 3D printing technology, which have made the production of customized implants and prosthetics more feasible and cost-effective. Overall, the global 3D Printing in Healthcare Market is anticipated to witness significant growth in the coming years, driven by the increasing adoption of 3D printing technology in the healthcare sector and the rising demand for personalized medical treatments. PEST Analysis: Political: The political factors impacting the 3D printing in healthcare market include government regulations and policies related to healthcare and medical devices. For example, the approval process for 3D printed medical devices may vary from country to country, which can affect the market growth. Additionally, government funding and support for research and development in the healthcare sector can also influence the adoption of 3D printing technology. Economic: The economic factors influencing the market include the cost-effectiveness of 3D printing in healthcare compared to traditional manufacturing methods. The growing demand for personalized healthcare solutions and the potential cost savings offered by 3D printing technology can drive market growth. Additionally, economic factors such as healthcare expenditure, GDP growth, and disposable income levels also play a role in the market's expansion. Social: The social factors impacting the market include the increasing awareness and acceptance of 3D printing in healthcare among both healthcare professionals and patients. The ability of 3D printing technology to customize medical devices and improve patient outcomes is driving its adoption. Additionally, the aging population and the rising prevalence of chronic diseases create a need for advanced and personalized medical solutions, which can be catered to by 3D printing technology. Technological: The technological factors influencing the market include advancements in 3D printing technology, such as the development of biocompatible materials and improved printing techniques. Additionally, the integration of 3D printing with other technologies like artificial intelligence and robotics can further enhance its capabilities in the healthcare sector. The ongoing research and development activities in the field are expected to contribute to the growth of the market. Key Takeaways: The global 3D Printing In Healthcare Market Forecast is projected to witness high growth, exhibiting a CAGR of 19.0% over the forecast period (2023-2030). This growth can be attributed to various factors, including the increasing demand for personalized healthcare solutions, the cost-effectiveness of 3D printing technology, and advancements in printing techniques and materials. In terms of regional analysis, North America is expected to be the fastest-growing and dominating region in the market. This can be attributed to factors such as the presence of major market players, strong healthcare infrastructure, and favorable government initiatives promoting the adoption of 3D printing in healthcare. Additionally, the Asia Pacific region is also expected to witness significant growth due to the increasing healthcare expenditure and technological advancements in countries like China and India. Key players operating in the 3D printing in healthcare market include 3D Systems Corporation, Exone Company, Formlabs Inc., General Electric (GE Additive), Materialise NV, Organovo Holdings, Inc., Oxford Performance Materials Inc., Proto Labs, Inc., Stratasys Ltd., and SLM Solutions Group AG. These companies are focusing on strategic collaborations, product launches, and technological advancements to strengthen their market position and expand their customer base. Read More - https://www.marketwebjournal.com/future-prospects-and-growth-opportunities-in-the-3d-printing-in-healthcare-market/
0 Comments
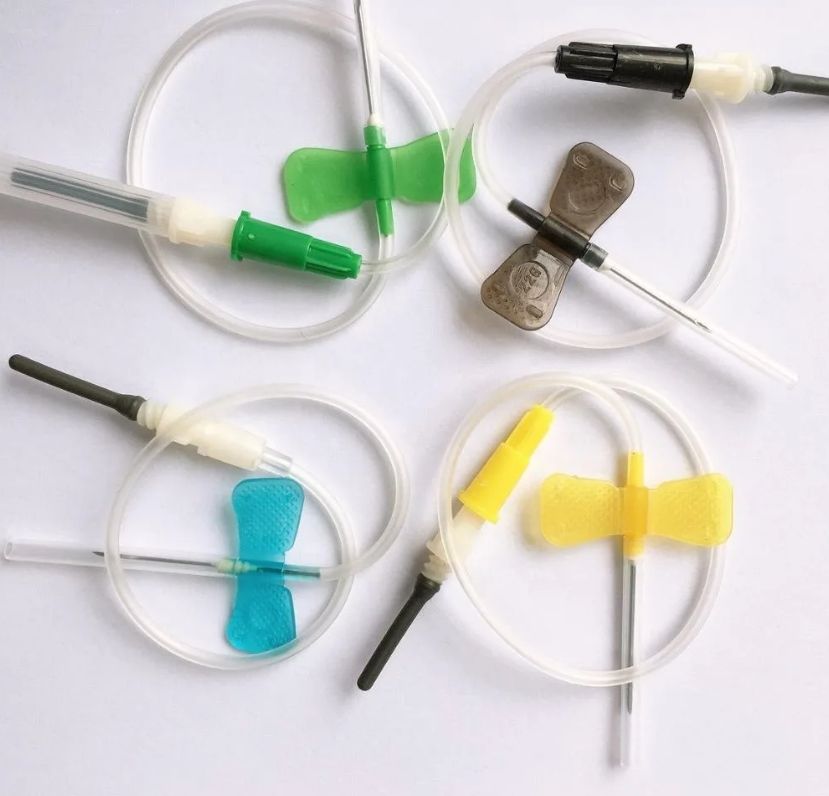
The global Butterfly Needles Market is estimated to be valued at US$639.4 million in 2023 and is expected to exhibit a CAGR of 9.8% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights.
Market Overview: Butterfly needles are medical devices used for venipuncture and blood collection. They are designed with a small winged-shaped handle, allowing for easy and secure handling during the procedure. Butterfly needles offer numerous advantages, including reduced pain and increased patient comfort, as well as improved vein visibility and easier blood flow. These devices are widely used in hospitals, clinics, diagnostic centers, and blood banks for various applications, such as blood transfusions, intravenous (IV) therapy, and collecting blood samples for laboratory testing. The increasing prevalence of chronic diseases, such as diabetes and cardiovascular diseases, is driving the demand for butterfly needles, as these diseases often require frequent blood tests and IV therapy. Market Key Trends: One key trend in the butterfly needles market is the growing adoption of safety butterfly needles. Safety butterfly needles are designed with safety features to reduce the risk of needlestick injuries. These needles have a built-in mechanism that activates after the procedure is completed, retracting the needle into a protective housing and preventing accidental needlestick injuries. The rising awareness about occupational hazards among healthcare professionals and the implementation of stringent regulations for workplace safety are driving the demand for safety butterfly needles. Additionally, the increasing prevalence of bloodborne diseases, such as HIV and Hepatitis, further emphasizes the need for safer medical devices, including butterfly needles. In conclusion, the global butterfly needles market is expected to experience significant growth due to the increasing prevalence of chronic diseases and the growing adoption of safety butterfly needles. The market is projected to witness high demand from hospitals, clinics, and diagnostic centers for various applications, driving PEST Analysis: Political: The political factors influencing the Butterfly Needles market include government regulations and policies regarding healthcare infrastructure, medical devices, and patient safety. For example, the approval process for introducing new medical devices in the market and the regulatory guidelines related to the manufacturing and distribution of Butterfly Needles can impact the growth of the market. Economic: Economic factors affecting the Butterfly Needles market include healthcare expenditure and reimbursement policies. The increasing healthcare expenditure and favorable reimbursement policies for medical procedures involving Butterfly Needles can drive market growth. Additionally, economic stability and disposable income of individuals also play a significant role in the adoption of advanced medical devices, including Butterfly Needles. Social: Social factors such as the aging population, prevalence of chronic diseases, and awareness about healthcare services influence the demand for Butterfly Needles. The rise in the geriatric population and the increasing prevalence of chronic illnesses, which require regular blood tests and intravenous therapies, drive the demand for Butterfly Needles. Technological: Technological advancements in Butterfly Needles contribute to market growth. Innovations in needle design, such as the development of safety needles and painless insertion techniques, improve patient experience and encourage the adoption of Butterfly Needles. Moreover, the integration of technology with these devices, such as electronic medical records and wireless connectivity, enhances the efficiency of healthcare delivery. Key Takeaways: The global Butterfly Needles Market Future is expected to witness high growth, exhibiting a CAGR of 9.8% over the forecast period (2023-2030). This growth can be attributed to factors such as increasing healthcare expenditure, favorable reimbursement policies, and technological advancements in the field. The market size is projected to reach US$ 639.4 million by 2023. In terms of regional analysis, North America is anticipated to be the fastest growing and dominating region in the Butterfly Needles market. The region's well-established healthcare infrastructure, high healthcare expenditure, and increasing prevalence of chronic diseases contribute to the market's growth. Additionally, the presence of key market players and their focus on research and development activities further boost the market in this region. Key players operating in the Butterfly Needles market include Chattanooga International, Humares, Prime Pacific Health, Herrmann Apparatebau, Transcom, CleanColon Italy, Prometheus Group, Shenzhen Lifotronic Technology, PPHIC, Comba UK, Enteromed Group, Lifotronic, Dotolo Research, and TECNO-GAZ. These companies are actively engaged in product development, strategic collaborations, and mergers and acquisitions to strengthen their market presence and expand their product portfolios. Read More - https://www.marketwebjournal.com/the-future-of-butterfly-needles-market-growing-demand-for-pain-free-vascular-access/ The global Bio Decontamination Market is estimated to be valued at US$ 178.3 Million in 2023 and is expected to exhibit a CAGR of 18.9% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights. Market Overview: Bio decontamination refers to the process of eliminating, neutralizing, or reducing harmful microorganisms from a surface, object, or environment. It is an essential step in ensuring sterilization and maintaining hygiene in various industries such as healthcare, pharmaceuticals, food processing, and research laboratories. Bio decontamination products play a crucial role in preventing the spread of infections and diseases, thereby ensuring the safety and well-being of individuals. Market Key Trends: One key trend in the bio decontamination market is the increasing adoption of advanced technologies for effective sterilization. With the growing concern for healthcare-associated infections and the need for stringent hygiene measures, there is a rising demand for innovative bio decontamination solutions. Advanced technologies such as hydrogen peroxide vapor (HPV) and chlorine dioxide gas are being increasingly used due to their superior efficacy in eliminating pathogens and reducing the risk of cross-contamination. These technologies offer advantages such as shorter decontamination cycles, compatibility with various materials, and ease of application, making them highly suitable for use in healthcare facilities, biopharmaceutical manufacturing, and research laboratories. The market is witnessing a shift towards automated bio decontamination systems, which offer improved efficiency and accuracy. These systems utilize sensors, robotics, and artificial intelligence to monitor and control the decontamination process, ensuring optimal results. Additionally, the integration of wireless connectivity and data analytics in bio decontamination equipment allows real-time tracking and analysis of sterilization processes, increasing reliability and quality assurance. The key players in the bio decontamination market include STERIS PEST Analysis: Political: The political factors influencing the bio decontamination market include regulations and policies implemented by governments regarding healthcare and safety standards. These policies can drive the demand for bio decontamination services in various industries such as pharmaceuticals, healthcare, food processing, and research laboratories. Economic: The economic factors affecting the bio decontamination market include the overall economic growth of countries, healthcare spending, and consumer disposable income. As economies grow, there is a higher demand for healthcare services, leading to an increased need for bio decontamination services. Social: The social factors influencing the bio decontamination market include the increasing awareness and concern for public health and safety. With a rise in infectious diseases and healthcare-associated infections, there is a growing need for effective bio decontamination solutions to prevent the spread of pathogens. Technological: The technological factors impacting the bio decontamination market include advancements in bio decontamination methods and equipment. Innovations in disinfection technologies, such as the use of hydrogen peroxide vapor or UV-C light, have led to more efficient and faster decontamination processes, driving market growth. Key Takeaways: The global Bio Decontamination Market Growth is expected to witness high growth, exhibiting a CAGR of 18.9% over the forecast period (2023-2030). This growth can be attributed to the increasing demand for bio decontamination services in various industries, including pharmaceuticals, healthcare, and food processing. The prevention and control of healthcare-associated infections are driving the adoption of bio decontamination solutions in healthcare facilities. Furthermore, the increasing focus on public health and safety, coupled with technological advancements in decontamination methods, is expected to further fuel market growth. In terms of regional analysis, North America is anticipated to be the fastest-growing and dominating region in the bio decontamination market. This can be attributed to the presence of established healthcare infrastructure, stringent regulations regarding infection control, and a high demand for bio decontamination services in industries such as pharmaceuticals and healthcare. The region is also home to key players in the bio decontamination market, contributing to its dominance in the market. Key players operating in the bio decontamination market include STERIS Corporation, Ecolab, Inc., TOMI Environmental Solutions, Inc., JCE Biotechnology, Fedegari Autoclavi SpA, Howorth Air Technology Ltd., Noxilizer, Inc., ClorDiSys Solutions, Inc., Wales Group, and Wenzhou Weike Biological Laboratory Equipment Co., Ltd. These market players offer a range of bio decontamination solutions and are involved in strategic initiatives such as collaborations, acquisitions, and product launches to gain a competitive edge in the market. Read More - https://www.marketwebjournal.com/bio-decontamination-market-is-estimated-to-witness-high-growth/ The global Cardiac Surgery Instrument Market is estimated to be valued at US$ 1.7 billion in 2023 and is expected to exhibit a CAGR of 13.0% over the forecast period 2023 to 2030, as highlighted in a new report published by Coherent Market Insights.
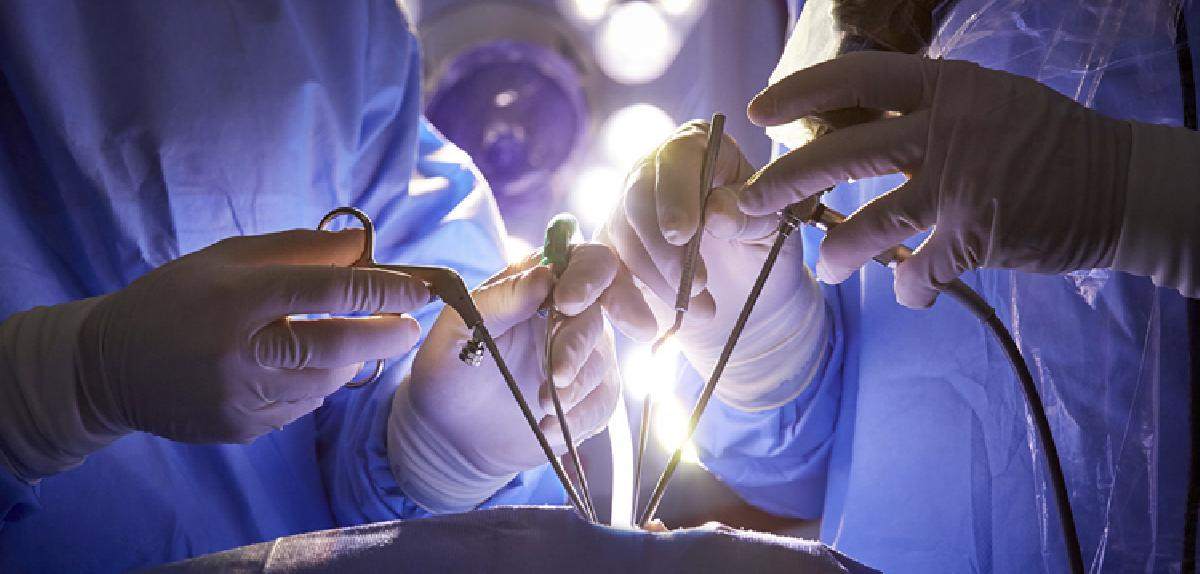
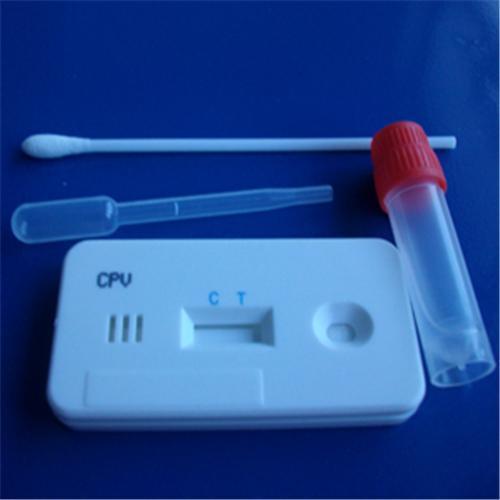
Market Overview: The cardiac surgery instrument market provides essential tools and equipment required for performing cardiac surgeries. These instruments play a crucial role in ensuring successful surgical procedures, improving patient outcomes, and enhancing overall surgical efficiency. With the increasing prevalence of cardiovascular diseases and the growing aging population, the demand for cardiac surgeries is rising significantly. The cardiac surgery instruments offer advantages such as precision, efficiency, and safety, which are necessary to perform complex procedures with optimal results. Hence, the market for cardiac surgery instruments is projected to experience high growth in the coming years. Market Key Trends: Technological advancements in cardiac surgery instruments are a key trend driving market growth. Innovative technologies such as robotic-assisted surgeries, minimally invasive procedures, and the development of advanced instruments for precise control and efficient surgical outcomes have revolutionized the field of cardiac surgery. These advanced instruments offer benefits such as reduced surgical trauma, shorter hospital stays, quicker recovery, and improved patient satisfaction. Furthermore, the integration of technologies such as artificial intelligence and virtual reality in cardiac surgery instruments is expected to further enhance surgical precision and outcomes. The increasing adoption of these advanced instruments and technologies by healthcare providers is anticipated to fuel market expansion in the forecast period. PEST Analysis: Political: The political factors influencing the cardiac surgery instrument market include government regulations and policies related to healthcare and medical devices. These regulations can impact the approval process for new surgical instruments and the overall market dynamics. Economic: Economic factors such as healthcare spending and reimbursement policies play a crucial role in the cardiac surgery instrument market. Increasing healthcare expenditure, particularly in emerging economies, is expected to drive market growth. The availability of insurance coverage and favorable reimbursement policies can also impact the demand for cardiac surgery instruments. Social: Social factors like the aging population, increasing prevalence of cardiovascular diseases, and growing awareness about advanced surgical procedures contribute to the growth of the market. The rising adoption of minimally invasive surgeries and technological advancements in cardiac surgery instruments also reflect the changing preferences and demands of patients. Technological: Rapid technological advancements, such as the development of robotic-assisted surgery and advanced imaging techniques, have significantly improved cardiac surgery procedures. Integration of artificial intelligence and data analytics in surgical instruments is expected to further enhance surgical outcomes and patient care. Continuous innovations in the field of cardiac surgery instruments drive market growth. Key Takeaways: The global Cardiac Surgery Instrument Market Forecast is expected to witness high growth, exhibiting a CAGR of 13.0% over the forecast period. This growth is driven by various factors, including the increasing prevalence of cardiovascular diseases and the rising adoption of minimally invasive surgical procedures. The market is also influenced by technological advancements in surgical instruments and an aging population. Regionally, North America is expected to be the fastest-growing and dominating region in the cardiac surgery instrument market. The region has well-established healthcare infrastructure, higher healthcare expenditure, and favorable reimbursement policies, which contribute to market growth. Furthermore, the presence of major market players and ongoing research and development activities support the growth of this region. Key players operating in the cardiac surgery instrument market include Medtronic, Edwards Lifesciences, Abbott Laboratories, Boston Scientific, LivaNova, Sorin Group (now part of LivaNova), Terumo Corporation, Getinge AB, CryoLife, MicroPort Scientific Corporation, B. Braun Melsungen AG, Maquet (now part of Getinge AB), W. L. Gore & Associates, Stryker Corporation, and Merit Medical Systems. These key players play a crucial role in driving market growth through product innovation, strategic partnerships, and mergers and acquisitions. The global Shigella Test Kit Market is estimated to be valued at US$ 106.4 million in 2023 and is expected to exhibit a CAGR of 5.9% over the forecast period (2023-2030), as highlighted in a new report published by Coherent Market Insights. Market Overview: Shigella test kits are diagnostic tools used to detect and identify the presence of the Shigella bacteria in stool samples. Shigella is a pathogenic bacterium that causes severe diarrheal diseases, and its detection is crucial for timely treatment and prevention of its spread. Shigella test kits offer several advantages, including rapid and accurate results, easy-to-use procedures, and cost-effectiveness. With the increasing prevalence of foodborne illnesses caused by Shigella bacteria, the demand for Shigella test kits is expected to rise significantly. Market Key Trends: One key trend driving the growth of the Shigella Test Kit Market is increasing awareness about foodborne illnesses. Governments and healthcare organizations worldwide are focusing on creating awareness about the risks associated with contaminated food and water. This has led to an increased demand for diagnostic tools such as Shigella test kits to identify and control foodborne diseases. For example, in 2019, the World Health Organization (WHO) launched a global strategy to reduce the burden of foodborne diseases, emphasizing the need for effective diagnostics. PEST Analysis: PEST Analysis for the Shigella Test Kit Market includes: 1. Political: Government regulations and policies regarding food safety and disease control affect the adoption of Shigella test kits. Stringent regulations for food manufacturers and healthcare facilities drive the demand for reliable diagnostic tools. 2. Economic: Increasing healthcare expenditure and rising awareness about food safety contribute to the growth of the Shigella test kit market. Economic stability and purchasing power also influence market growth. 3. Social: Growing concern about food safety among consumers and healthcare professionals drives the demand for Shigella test kits. The increasing prevalence of foodborne diseases and the need for prompt diagnosis fuel market growth. 4. Technological: Advancements in diagnostic technologies, such as polymerase chain reaction (PCR) and enzyme-linked immunosorbent assays (ELISA), have improved the accuracy and efficiency of Shigella test kits. Technological advancements play a significant role in driving market growth. Key Takeaways: - The global Shigella Test Kit Market is expected to witness high growth, exhibiting a CAGR of 5.9% over the forecast period, due to increasing awareness about foodborne illnesses. - In terms of regional analysis, North America is anticipated to be the fastest-growing and dominating region in the market. The presence of major key players, well-established healthcare infrastructure, and strict food safety regulations contribute to the region's market growth. - Key players operating in the global Shigella Test Kit Market include Thermo Fisher Scientific Inc., Meridian Bioscience Inc., QIAGEN N.V., Bio-Rad Laboratories Inc., Creative Diagnostics, Hardy Diagnostics, Luminex Corporation, DiaSorin S.p.A., Sekisui Diagnostics LLC, BioMérieux SA, MP Biomedicals LLC, Coris BioConcept, Alere Inc., Fujirebio Diagnostics Inc., and CTK Biotech Inc. These players focus on product development, strategic collaborations, and mergers and acquisitions to strengthen their market position. In conclusion, the global Shigella Test Kit Market is poised to experience significant growth due to increasing awareness about foodborne illnesses and the need for accurate diagnostic tools. Governments' initiatives and investments in food safety and disease control further contribute to the market's expansion. North America is expected to dominate the market, while key players continue to innovate and collaborate to stay competitive in the market. The global Medical Devices Reimbursement Market is estimated to be valued at US$ 501.8 Mn in 2022 and is expected to exhibit a CAGR of 8.6% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights. A) Market Overview: Medical devices reimbursement refers to the process of receiving payment for medical devices and equipment from insurance providers or government healthcare programs. The market for medical devices reimbursement is driven by the increasing penetration of health insurance policies across the globe. As healthcare costs continue to rise, individuals are seeking financial protection through health insurance plans that cover medical devices and equipment. This creates a favorable environment for the growth of the medical devices reimbursement market. B) Market Key Trends: One key trend in the medical devices reimbursement market is the growing demand for innovative and advanced medical devices. With advancements in technology, medical devices are becoming more sophisticated, leading to improved patient outcomes and reduced healthcare costs. For example, the introduction of minimally invasive surgical devices has revolutionized surgical procedures by reducing hospital stays and recovery time. This trend is driving the adoption of such innovative medical devices, which in turn fuels the growth of the medical devices reimbursement market. C) PEST Analysis: - Political: The political landscape plays a significant role in shaping the medical devices reimbursement market. Government policies and regulations related to healthcare funding and insurance coverage can impact reimbursement rates and accessibility to medical devices. - Economic: Economic factors like GDP growth, healthcare expenditure, and insurance coverage rates influence the demand for medical devices reimbursement. A growing economy with increased healthcare spending capacity can drive market growth. - Social: Social factors such as aging populations, increasing chronic diseases, and rising awareness about healthcare contribute to the growing demand for medical devices reimbursement. - Technological: Technological advancements in medical devices and equipment drive the need for reimbursement. Newer and more effective devices often come at a higher cost, necessitating coverage from insurance providers. D) Key Takeaways: In terms of market size, the global medical devices reimbursement market is expected to witness high growth, exhibiting a CAGR of 8.6% over the forecast period. This growth is mainly driven by increasing penetration of health insurance policies that cover medical devices and equipment. Regionally, North America is expected to dominate the medical devices reimbursement market, owing to the presence of key players such as BNP Paribas, CVS Health, and UnitedHealth Group Inc. Additionally, the region has a well-established healthcare insurance system and a high penetration rate of health insurance policies. Key players operating in the global Medical Device Reimbursement Market include BNP Paribas, CVS Health, Aviva, Allianz, Humana, Cigna, Aetna, Wellcare Health Plans Inc., UnitedHealth Group Inc., and Nippon Life Insurance Company. These players are actively involved in providing reimbursement services for medical devices and equipment. In conclusion, the global market for medical devices reimbursement is expected to witness significant growth in the coming years. The increasing adoption of health insurance policies and the demand for innovative medical devices are the key drivers for this growth. North America is anticipated to dominate the market, and key players such as BNP Paribas and CVS Health play a crucial role in providing reimbursement services. The global In Silico Clinical Trials Market is estimated to be valued at US$ 3,173.1 Mn in 2022 and is expected to exhibit a CAGR of 7.95% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights.
A) Market Overview: Silico Clinical Trials are virtual trials conducted using computer models and simulations to evaluate drug efficacy and safety. These trials provide several advantages over traditional clinical trials, such as reduced time and cost, ethical considerations, and the ability to collect large amounts of data. Virtual trials eliminate the need for physical participation, making it more convenient for patients and reducing the burden on healthcare systems. They also allow for the exploration of various drug combinations and dosages, accelerating the development of personalized medicine. B) Market Key Trends: One key trend in the In Silico Clinical Trials Market is the growing demand for virtual clinical trials. With advancements in technology and the increasing need for efficient drug development processes, pharmaceutical companies are increasingly adopting virtual trials. These trials provide real-time data analysis, reduce patient recruitment time, and offer cost-effective solutions for drug development. For example, Insilico Medicine Inc., a key player in the market, utilizes artificial intelligence (AI) algorithms to accelerate the drug discovery process. Their AI-based platform enables researchers to identify potential drug candidates and predict their efficacy using virtual models. C) PEST Analysis: Political: Regulatory frameworks play a crucial role in the adoption of virtual clinical trials. Governments need to establish guidelines and standards to ensure patient safety and data privacy. Economic: Silico Clinical Trials offer cost-effective solutions compared to traditional clinical trials. They reduce the need for physical locations, extensive patient recruitment efforts, and travel expenses. Social: Virtual trials provide opportunities for patients who may otherwise be unable to participate in traditional trials due to geographical constraints, physical limitations, or personal commitments. Technological: Advancements in technology, such as AI, machine learning, and big data analytics, have facilitated the growth of virtual trials. These technologies enable researchers to analyze complex data sets and make predictions about drug efficacy and safety. D) Key Takeaways: Paragraph 1: The global In Silico Clinical Trials Market is expected to witness high growth, exhibiting a CAGR of 7.95% over the forecast period. This growth can be attributed to increasing demand for virtual trials due to their ability to reduce time and cost in drug development. For example, the use of virtual models and simulations enables researchers to predict drug efficacy and safety before conducting physical trials. Paragraph 2: Regionally, North America is expected to dominate the In Silico Clinical Trials Market . The region has a well-established healthcare infrastructure, favorable regulatory environment, and a high adoption rate of advanced technologies. Additionally, collaborations between pharmaceutical companies, academic institutions, and research organizations in North America contribute to the region's growth in the market. Paragraph 3: Key players operating in the global In Silico Clinical Trials Market include Abzena Ltd., Clarivate, Immunetrics Inc., GNS Healthcare, Dassault Systemes, Evotec, Novadiscovery, Insilico Medicine Inc., and InSilicoTrials Technologies. These companies are actively involved in research and development activities to enhance their virtual trial capabilities and offer innovative solutions to pharmaceutical companies. In conclusion, the In Silico Clinical Trials Market is witnessing significant growth due to the increasing demand for virtual trials. Advancements in technology, cost-effectiveness, and the ability to collect real-time data are driving the adoption of virtual trials in drug development. The market is dominated by key players who are continuously investing in research and development to stay ahead in the competitive landscape. As virtual trials become more widely accepted, they have the potential to revolutionize the drug development process and improve patient outcomes. The global Drug Eluting Balloon Market is estimated to be valued at US$ 616.6 million in 2022 and is expected to exhibit a CAGR of 8.3% over the forecast period 2022-2030, as highlighted in a new report published by Coherent Market Insights.
A) Market Overview: Drug eluting balloons are medical devices used in interventional cardiology procedures to deliver drugs directly to the targeted lesion. These balloons are particularly effective in preventing artery re-narrowing after angioplasty and stent placement. The advantages of drug eluting balloons include reduced rates of restenosis, decreased need for repeat procedures, and improved patient outcomes. The increasing prevalence of cardiovascular diseases, such as coronary artery disease and peripheral arterial disease, is driving the demand for drug eluting balloons. B) Market Key Trends: One key trend in the drug eluting balloon market is the development of advanced coatings for drug eluting balloons. Manufacturers are investing in research and development activities to improve the properties and efficacy of drug delivery systems. For instance, Medtronic developed the IN.PACT Admiral drug eluting balloon with a unique coating called FreePac, which provides controlled drug release and reduces inflammation. This coating helps in minimizing complications and improving patient outcomes. C) PEST Analysis: Political: The political landscape plays a crucial role in the Drug Eluting Balloon Market, as government regulations and policies impact product approvals and market access. Economic: Economic factors, such as healthcare expenditure and reimbursement policies, influence the adoption of drug eluting balloons. Social: The increasing elderly population and lifestyle changes leading to a higher incidence of cardiovascular diseases are social factors driving the market. Technological: Advancements in technology, such as the development of innovative coatings and drug delivery systems, are shaping the drug eluting balloon market. D) Key Takeaways: - The global drug eluting balloon market is expected to witness high growth, exhibiting a CAGR of 8.3% over the forecast period, due to increasing prevalence of cardiovascular diseases and advancements in technology. - Regionally, North America is the fastest-growing and dominating region in the drug eluting balloon market, due to a high burden of cardiovascular diseases and well-established healthcare infrastructure. - Key players operating in the global drug eluting balloon market include Medtronic, Becton, Dickinson and Company (BD), Boston Scientific Corporation, Cook Medical, B. Braun Melsungen AG, Koninklijke Philips N.V., Opto Circuits (India) Limited, Terumo Corporation, Surmodics, Inc., and Biotronik. In conclusion, the global drug eluting balloon market is experiencing significant growth due to the increasing prevalence of cardiovascular diseases. Advancements in technology, such as advanced coatings for drug eluting balloons, are further driving market growth. North America is the fastest-growing region in the market, dominated by key players such as Medtronic and Boston Scientific Corporation. With the rising demand for effective treatment options for cardiovascular diseases, the drug eluting balloon market is expected to witness sustained growth in the coming years. The global Refurbished Medical Equipment Market is estimated to be valued at USD 9.9 billion in 2022 and is expected to exhibit a CAGR of 11.6% over the forecast period of 2023-2032, as highlighted in a new report published by Coherent Market Insights. The growing need for cost-effective medical devices, coupled with the increasing focus on sustainable and green healthcare solutions, is driving the demand for refurbished medical equipment in the market.
A) Market Overview: The Refurbished Medical Equipment Market comprises a range of medical devices that have been restored to their original condition or upgraded to meet current standards. These devices offer an affordable alternative to new equipment, making healthcare services more accessible, especially in resource-constrained settings. The market includes refurbished imaging equipment, surgical instruments, patient monitoring devices, and others. B) Market Dynamics: 1. Cost-effectiveness: The rising healthcare costs and budget constraints faced by healthcare facilities have led to a growing demand for refurbished medical equipment. These devices are available at a significantly lower cost compared to new equipment, enabling healthcare providers to deliver quality care while optimizing their resources. 2. Focus on sustainability: The market is witnessing a shift towards sustainable and green healthcare solutions. Refurbished medical equipment promotes the reuse and recycle of products, reducing e-waste and environmental impact. This factor is further driving the adoption of refurbished equipment among healthcare providers. C) Market Key Trends: One key trend in the Refurbished Medical Equipment Market is the increasing demand for refurbished imaging equipment. Imaging devices such as MRI machines, CT scanners, and ultrasound systems are expensive to purchase and maintain. Refurbished imaging equipment offers a cost-effective solution for healthcare facilities to upgrade their imaging capabilities without a substantial investment. For example, hospitals in emerging economies are increasingly opting for refurbished imaging equipment to enhance their diagnostic capabilities at a lower cost. D) SWOT Analysis: - Strengths: 1. Cost-effectiveness: Refurbished medical equipment offers significant cost savings compared to new equipment. 2. Sustainability: The reuse and recycle of refurbished equipment reduce e-waste and environmental impact. - Weaknesses: 1. Limited warranty and support: Refurbished equipment may have a shorter warranty period and limited support compared to new equipment. 2. Potential quality concerns: There may be concerns related to the quality and reliability of refurbished equipment. - Opportunities: 1. Growing demand in emerging economies: The demand for cost-effective healthcare solutions in emerging economies presents significant growth opportunities for the refurbished medical equipment market. 2. Technological advancements: Advancements in refurbishment processes and technologies can improve the quality and reliability of refurbished equipment. - Threats: 1. Stringent regulations: Regulatory requirements may pose challenges for the sale and distribution of refurbished medical equipment. 2. Competition from low-cost alternatives: Low-cost alternatives, such as counterfeit or unregulated devices, can pose a threat to the market. E) Key Takeaways: - The global Refurbished Medical Equipment Market is expected to witness high growth, exhibiting a CAGR of 11.6% over the forecast period, due to the increasing demand for cost-effective medical devices. - North America is the fastest-growing and dominating region in the market, driven by advanced healthcare infrastructure, high healthcare expenditure, and the presence of key players. - Key players operating in the global Refurbished Medical Equipment Market include GE Healthcare, Philips Healthcare, Siemens Healthineers, Soma Technology, Inc., Block Imaging International, Inc., and Agito Medical A/S. These players are focused on strategies such as partnerships, collaborations, and acquisitions to expand their market presence. In conclusion, the Refurbished Medical Equipment Market is witnessing significant growth due to the rising demand for cost-effective healthcare solutions and a focus on sustainability. The market offers opportunities for healthcare providers to upgrade their equipment at a lower cost while contributing to environmental conservation. However, regulatory challenges and potential quality concerns remain key considerations for market players. In the realm of medical diagnostics, the Colonoscopy Devices Market has emerged as a pivotal player, transforming the landscape of gastrointestinal health assessments. With the growing prevalence of colorectal diseases and the rising adoption of preventive healthcare, this market is undergoing rapid innovation, propelling it to new heights of efficacy and patient comfort.
The Colonoscopy Devices Market is witnessing a paradigm shift, fueled by technological advancements that are redefining the diagnostic experience. Traditional colonoscopy procedures were often met with anxiety and discomfort due to the invasive nature of the examination. However, modern colonoscopy devices are integrating high-definition imaging, ultra-slim designs, and advanced maneuverability, ensuring a less intrusive and more accurate assessment. One of the groundbreaking trends in this market is the incorporation of artificial intelligence (AI) and machine learning. These technologies empower colonoscopists with real-time analysis and insights, enhancing the detection of polyps and abnormalities. AI-driven colonoscopy devices are poised to reduce human error, improve diagnostic accuracy, and ultimately save lives through early detection of colorectal diseases. Moreover, the Colonoscopy Devices Market is experiencing a surge in the development of disposable and single-use colonoscopy devices. These innovative solutions eliminate the need for meticulous cleaning and sterilization processes, ensuring patient safety and reducing the risk of cross-contamination. As healthcare institutions prioritize infection control, these devices are gaining traction for their convenience and hygienic benefits. Telemedicine is another pivotal trend shaping the Colonoscopy Devices Market. With the advent of remote medical consultations, patients can now undergo colonoscopy procedures while being remotely monitored by healthcare professionals. This approach not only enhances accessibility, especially in remote areas, but also minimizes the need for patients to travel long distances for screenings. As patient comfort takes center stage, colonoscopy devices are being designed with features that alleviate discomfort and anxiety. Automated insertion and withdrawal technologies, along with advanced sedation techniques, are contributing to a more relaxed patient experience. These innovations are expected to drive higher participation rates in colorectal screenings, thereby leading to earlier disease detection. In conclusion, the Colonoscopy Devices Market is on a transformative trajectory, fueled by technological innovations that prioritize accuracy, patient comfort, and convenience. The integration of AI, disposable devices, and telemedicine solutions are redefining the landscape of colonoscopy procedures, making them less invasive, more accurate, and accessible to a wider population. As the market continues to evolve, individuals can look forward to enhanced gastrointestinal health assessments that have the potential to revolutionize early disease detection and overall patient well-being. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|



 RSS Feed
RSS Feed
