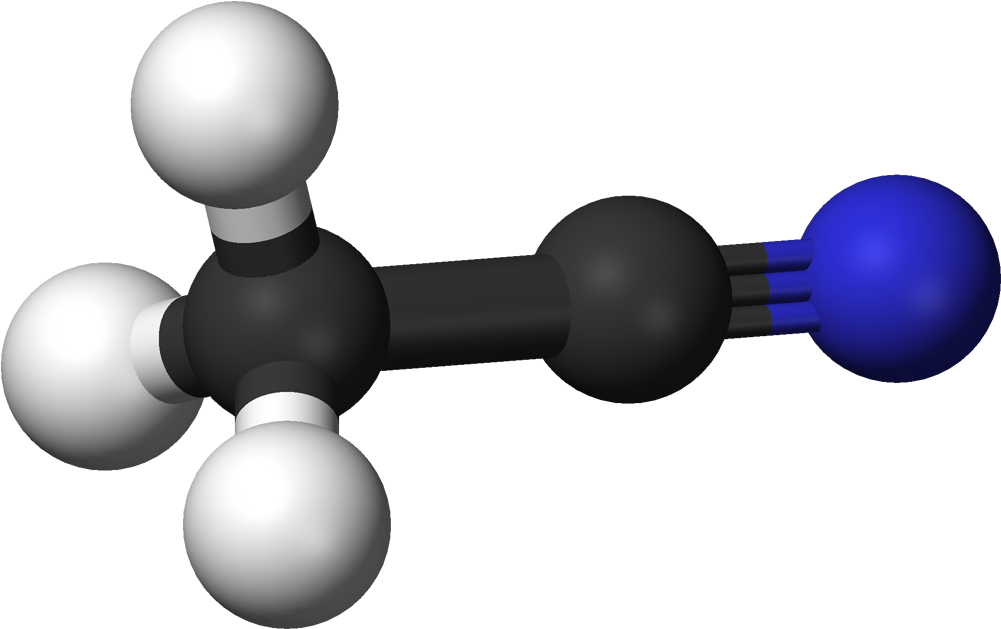
Acetonitrile Is A Nitrile Composed Of Hydrogen Cyanide With A Methyl Group In Lieu Of The Hydrogen4/24/2023 Acetonitrile functions as a polar aprotic solvent as well as an EC 3.5.1.4 (amidase) inhibitor. It is a volatile organic chemical and an aliphatic nitrile. The chemical molecule Acetonitrile, often known as MeCN (methyl cyanide), has the formula CH3CN and the structural formula H3CCN.
The simplest organic nitrile is this colourless liquid; hydrogen cyanide is another simple nitrile, but the cyanide anion is not considered to be organic. It is mostly created as a by-product of the production of acrylonitrile. It serves as a polar aprotic solvent for chemical synthesis and butadiene purification. The CN distance of the NCC skeleton is only 1.16, making it linear. Acetonitrile is primarily used as a solvent in refineries to purify butadiene. Acetonitrile is specifically injected into the top of a distillation column that contains butadiene and other hydrocarbons, and as it descends through the column, it absorbs the butadiene, which is then delivered from the bottom of the tower to a second separate tower. The butadiene is then separated using heat in the separating tower. It is employed as a medium-polarity solvent in the lab and is miscible with water and a variety of organic solvents, but not saturated hydrocarbons. Its high dielectric constant of 38.8 and convenient liquid range are both advantages. Acetonitrile, which has a dipole moment of 3.92 D, is a useful mobile phase in HPLC and LC-MS and dissolves a variety of ionic and nonpolar molecules. Due to its relatively high dielectric constant and capacity to dissolve electrolytes, it is frequently employed in battery applications. It is a common solvent in cyclic voltammetry for similar reasons. For high-performance liquid chromatography (HPLC), its UV cut-off, low viscosity, and minimal chemical reactivity make it a desirable option. As the primary solvent employed in the synthesis of oligonucleotides from nucleoside phosphoramidites, Acetonitrile plays a key role. In the manufacturing of medications and photographic film, it serves as a solvent. In the organic synthesis of several important compounds, such as acetamidine hydrochloride, thiamine, and -napthaleneacetic acid, Acetonitrile is a frequent two-carbon building block. Malononitrile is produced when it interacts with cyanogen chloride
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|
 RSS Feed
RSS Feed
