|
Tissue engineering is a ground-breaking field that combines biology, engineering, and medicine to create functional tissues and organs. This innovative approach holds immense promise for regenerative medicine, offering hope for patients with organ failure, tissue damage, or congenital abnormalities.
In this article, we will explore the concept of Tissue Engineering, discuss its applications, highlight its benefits and challenges, and delve into its potential to transform healthcare. Understanding Tissue Engineering Tissue Engineering involves the creation of artificial tissues or organs by combining cells, biomaterials, and biochemical factors. Scientists cultivate cells in a lab and provide them with an appropriate environment to grow and differentiate into specific tissue types. Scaffold materials, such as biodegradable polymers, provide structural support and guide cell growth. Bioreactors and specialized culture techniques help mimic the physiological conditions necessary for tissue development. The ultimate goal is to create functional tissues that can integrate seamlessly with the patient's body, restoring form and function. Applications of Tissue Engineering Tissue engineering has a wide range of applications in regenerative medicine. It has the potential to revolutionize the treatment of various conditions, including: Organ Transplantation: The shortage of donor organs poses a significant challenge in healthcare. Tissue engineering offers the possibility of growing organs in the lab, reducing the need for organ transplantation and eliminating the risk of organ rejection. Wound Healing: Tissue-engineered skin substitutes can promote wound healing and aid in the regeneration of damaged tissues. They provide a natural environment for cell growth and accelerate the healing process. Cartilage and Bone Repair: Tissue-engineered constructs can be used to regenerate damaged cartilage and bone tissues, providing a promising alternative to traditional joint replacements and bone grafts. Cardiac Tissue Repair: Tissue engineering holds potential for regenerating damaged heart tissue after a heart attack. Bioengineered cardiac patches or 3D-printed scaffolds can help restore cardiac function. Benefits and Challenges Tissue engineering offers several advantages over traditional treatments: Personalized Medicine: Tissue-engineered constructs can be customized to match the patient's specific needs, increasing the likelihood of successful integration and reducing the risk of rejection. Reduced Dependency on Donor Organs: By creating organs in the lab, tissue engineering addresses the shortage of donor organs and eliminates the need for immunosuppressive drugs. Improved Quality of Life: Tissue engineering aims to restore functionality and improve the quality of life for patients with tissue damage or organ failure. However, there are challenges to overcome: Complex Tissue Structure: Replicating the intricate structure and functionality of native tissues is a complex task that requires advances in biomaterials, cell culturing techniques, and tissue integration. Vascularization: Creating a network of blood vessels within tissue-engineered constructs is crucial for their long-term survival. Overcoming the challenges of vascularization remains a focus of ongoing research. Regulatory and Ethical Considerations: The development and approval of tissue-engineered products require adherence to regulatory guidelines and ethical considerations, ensuring their safety and efficacy. Increasing prevalence of obesity is expected to boost growth of the global Soft Tissue Repair Market over the forecast period. The Future of Tissue Engineering Tissue engineering holds tremendous potential for the future of healthcare. Ongoing research and technological advancements are driving the field forward. Scientists are exploring new biomaterials, developing advanced 3D printing techniques, and improving the understanding of cell signaling and tissue development. The integration of tissue engineering with other emerging technologies, such as stem cell therapy and gene editing, offers exciting possibilities for enhancing regenerative medicine approaches. With continued advancements and collaboration between scientists, engineers, and clinicians, Tissue Engineering has the potential to transform healthcare by providing innovative solutions for tissue repair, organ replacement, and personalized regenerative therapies. Increasing funding in the life science sector is expected to boost growth of the global Life Science Products Market over the forecast period. Tissue engineering is a dynamic and rapidly evolving field that brings together the realms of biology, engineering, and medicine. It holds immense promise for regenerative medicine, offering solutions to the challenges of organ transplantation, tissue repair, and functional restoration. While there are still challenges to overcome, the future of Tissue Engineering looks promising. As scientists continue to push the boundaries of scientific knowledge and technological capabilities, tissue engineering has the potential to revolutionize healthcare, improve patient outcomes, and pave the way for a new era of regenerative medicine. Know More - https://www.prnewswire.com/news-releases/global-tissue-engineering-market-to-surpass-us-22-908-1-million-by-2030--coherent-market-insights-301602281.html
0 Comments
In the field of biotechnology, small scale bioreactors have emerged as powerful tools for conducting research and development activities. These compact and versatile devices provide a controlled environment for the growth and study of various biological systems, such as cells, microorganisms, and tissues. With their ability to mimic the conditions found in larger industrial bioreactors, small scale bioreactors offer numerous advantages, making them indispensable in various areas of biotechnology research. One of the key advantages of Small Scale Bioreactors is their ability to provide a cost-effective solution for conducting experiments. Traditional large-scale bioreactors require significant resources, space, and capital investment. In contrast, small scale bioreactors are compact and can be easily set up in a laboratory or research facility. This allows researchers to perform experiments on a smaller scale, reducing the amount of costly reagents and samples required. Additionally, small scale bioreactors enable researchers to optimize experimental conditions and parameters before scaling up to larger production processes, minimizing the risk of costly failures. Another significant benefit of Small Scale Bioreactors is their flexibility and scalability. Researchers can customize the design and parameters of the bioreactor to suit their specific research needs. This adaptability allows for the cultivation of various cell types, including mammalian cells, microbial cultures, and plant cells. Small scale bioreactors can be easily modified to simulate specific physiological conditions, such as pH, temperature, oxygen levels, and nutrient availability, providing a controlled environment for studying cell behavior and metabolic processes. Furthermore, the data obtained from small scale bioreactors can be extrapolated and used to optimize larger-scale bioprocesses, facilitating the translation of research findings into industrial applications. The integration of advanced monitoring and control systems in Small Scale Bioreactors has revolutionized the way researchers conduct experiments. These bioreactors are equipped with sensors and automated control mechanisms that continuously monitor and adjust critical parameters, ensuring optimal growth conditions for the cells or microorganisms being cultured. Cell Expansion Market is expected to grow as there is increasing R&D activities for the use of cell therapy technique for the development of vaccines, antibiotics, and therapeutics by the key market players. Real-time data acquisition allows researchers to closely monitor cell viability, growth rates, nutrient consumption, and metabolite production. The ability to control and manipulate these variables enhances reproducibility and facilitates the study of complex biological processes. Small Scale Bioreactors have also opened new avenues for research in emerging fields such as tissue engineering and regenerative medicine. By providing a controlled environment for the growth and differentiation of stem cells, small scale bioreactors enable researchers to study tissue formation and develop innovative strategies for tissue repair and replacement. The use of small scale bioreactors in tissue engineering has the potential to revolutionize the field, bringing us closer to the day when complex organs can be engineered in the laboratory for transplantation. In conclusion, Small Scale Bioreactors have become invaluable tools in the field of biotechnology research. Their cost-effectiveness, flexibility, scalability, and advanced monitoring capabilities make them ideal for conducting a wide range of experiments. Global m-RNA Synthesis Service Market growth is expected to propel over the forecast period, owing to rising government initiatives and efforts for the development of mRNA therapeutics, especially during the emergence of the COVID-19 pandemic. By enabling researchers to optimize experimental conditions and gather valuable data, small scale bioreactors contribute to the advancement of biotechnology and the development of innovative solutions in areas such as pharmaceuticals, biofuels, and personalized medicine. As technology continues to evolve, small scale bioreactors will undoubtedly play a pivotal role in shaping the future of biotechnology research. Top of Form In the realm of medical advancements, few breakthroughs have been as promising and transformative as Cell Therapy. This cutting-edge treatment has captured the attention of scientists, healthcare professionals, and patients alike, heralding a new era of hope and healing. By harnessing the power of cells, Cell Therapy is revolutionizing the way we approach disease treatment and regeneration. At its core, Cell Therapy involves the utilization of living cells to restore, repair, or replace damaged tissues and organs within the body. This remarkable approach holds immense potential across a wide spectrum of medical conditions, ranging from cancer to autoimmune disorders, neurodegenerative diseases, and even tissue injuries. By tapping into the body's innate regenerative abilities, Cell Therapy aims to unlock its full potential for healing. One of the key aspects of Cell Therapy lies in the utilization of stem cells. These remarkable cells have the extraordinary ability to differentiate into various specialized cell types, making them ideal candidates for tissue regeneration. Stem cells can be sourced from various places, such as bone marrow, adipose tissue, or even umbilical cord blood. By isolating and culturing these cells in a controlled laboratory environment, scientists can generate an abundant supply of therapeutic cells for transplantation. Cell Therapy holds immense promise in the field of cancer treatment. Traditional cancer therapies like chemotherapy and radiation often come with significant side effects, as they not only target cancer cells but also harm healthy cells in the process. Cell Therapy, on the other hand, offers a more targeted approach. By engineering immune cells or introducing therapeutic genes into the patient's own cells, Cell Therapy becomes a personalized treatment option that specifically targets cancer cells, leaving healthy cells unharmed. Moreover, Cell Therapy can also play a pivotal role in addressing autoimmune disorders. In conditions like rheumatoid arthritis or multiple sclerosis, the body's immune system mistakenly attacks its own tissues, leading to chronic inflammation and tissue damage. By modulating the immune response with Cell Therapy, researchers aim to restore balance and prevent further damage. This approach shows great promise in alleviating symptoms and even achieving disease remission. Neurodegenerative diseases, such as Alzheimer's and Parkinson's, have long posed significant challenges to the medical community. However, Cell Therapy offers a glimmer of hope in this domain as well. By transplanting neural stem cells or promoting the growth and differentiation of existing brain cells, researchers aim to repair damaged neural circuits and restore cognitive function. While still in the early stages of development, these therapies hold immense potential for tackling these devastating conditions. Cell Therapy potential is not limited to complex diseases alone; it also shows promise in treating tissue injuries and promoting regeneration. Whether it's a sports injury or a severe burn, the ability to replace damaged tissues with healthy cells opens up a world of possibilities for faster and more complete recoveries. By enhancing the body's natural healing processes through Cell Therapy, patients may experience improved outcomes and reduced scarring. In conclusion, Cell Therapy stands as a healing marvel that holds the key to unlocking the body's full regenerative potential. With its ability to repair, replace, and regenerate tissues and organs, it offers hope to millions of people around the world battling various diseases and injuries. Biopharmaceuticals are medicinal products derived from living organisms, including proteins, antibodies, and nucleic acids, used for therapeutic purposes. Whether it's harnessing the power of stem cells or engineering immune cells, Cell Therapy's potential is vast and continues to be explored by researchers worldwide. As science and technology advance, we can expect Cell Therapy to play an increasingly significant role in revolutionizing medicine and improving the lives of countless individuals. Read More- https://www.globenewswire.com/news-release/2023/01/12/2587958/0/en/Global-Cell-Therapy-Market-to-Surpass-US-1-391-94-Million-by-2030-Says-Coherent-Market-Insights-CMI.html In the field of bioengineering, the advent of small scale bioreactors has sparked a revolution in research and development. These compact yet powerful devices are propelling bioengineering innovations to new heights, unlocking possibilities that were once unimaginable.
Small Scale Bioreactors have emerged as a cornerstone of bioengineering research, enabling scientists to explore and manipulate biological systems on a miniature scale. These innovative tools replicate the conditions found in larger bioreactors, but with the advantage of reduced volumes and increased flexibility. This miniaturization allows for rapid experimentation, cost savings, and greater control over experimental parameters. The impact of Small Scale Bioreactors on bioengineering research is far-reaching. Their versatility and scalability make them ideal for a wide range of applications, from studying cellular behavior to developing new therapeutic treatments. By simulating the complex biological processes in a controlled environment, scientists can gain valuable insights into the mechanisms of disease, drug efficacy, and tissue regeneration. One of the key advantages of small scale bioreactors is their ability to accelerate the pace of innovation. With their compact size, researchers can simultaneously run multiple experiments, optimizing time and resources. This parallel experimentation allows for faster data generation, hypothesis testing, and iterative design improvements. The rapid feedback loop facilitated by small scale bioreactors accelerates the discovery and development of novel bioengineering solutions. Moreover, Small Scale Bioreactors offer significant cost savings compared to traditional large-scale bioreactors. The reduced footprint and lower consumption of reagents, media, and energy contribute to a more cost-effective research process. This cost efficiency enables more laboratories and research institutions to access and utilize bioreactor technology, fostering collaboration and driving collective advancements in the field. Small scale bioreactors are particularly transformative in the emerging field of personalized medicine. The ability to culture patient-specific cells in miniature bioreactors opens up new avenues for tailored treatments and precision medicine. By mimicking the physiological conditions of individual patients, small scale bioreactors allow for the testing of personalized therapies, ultimately improving patient outcomes and reducing healthcare costs. The rise of miniaturization in bioreactors also addresses key challenges in bioengineering, such as scalability and translatability. Large-scale production of biopharmaceuticals often requires the transition from small scale research to industrial-scale manufacturing. Small Scale Bioreactors serve as a bridge, allowing researchers to develop robust processes that can be easily scaled up for commercial production. This seamless transition from lab-scale to large-scale bioreactors enhances the efficiency and reliability of bio manufacturing, bringing innovative therapies to patients more quickly. The utilization of small scale bioreactors is not without its considerations. The intricacies of miniaturization require careful calibration and optimization of parameters to ensure reproducibility and reliability. The culture conditions, nutrient supply, and bioprocess monitoring must be meticulously controlled to achieve consistent and meaningful results. However, the advancements in automation and real-time monitoring systems have significantly enhanced the capabilities and ease of use of small scale bioreactors, mitigating potential challenges. In conclusion, the rise of miniaturization in the form of Small Scale Bioreactors has revolutionized bioengineering research and development. These compact devices have become indispensable tools in the pursuit of scientific discoveries, therapeutic advancements, and personalized medicine. Cell And Gene Therapy plays a major role in modern healthcare, as it provides concepts and techniques that can be used in gene regulation, cell-cell interactions, regenerative capacity, and remodeling. With their ability to accelerate innovation, improve cost efficiency, and bridge the gap between research and commercialization, Small Scale Bioreactors are propelling bioengineering innovations to unprecedented levels. As the field continues to evolve, we can expect even more remarkable breakthroughs driven by the power of small scale bioreactors. In a ground-breaking development, the medical community has witnessed a significant breakthrough in the realm of emergency response to venomous bites. The superhero of this narrative is none other than anti-venom, a remarkable medical innovation that has revolutionized the way we combat the deadly effects of venomous creatures.
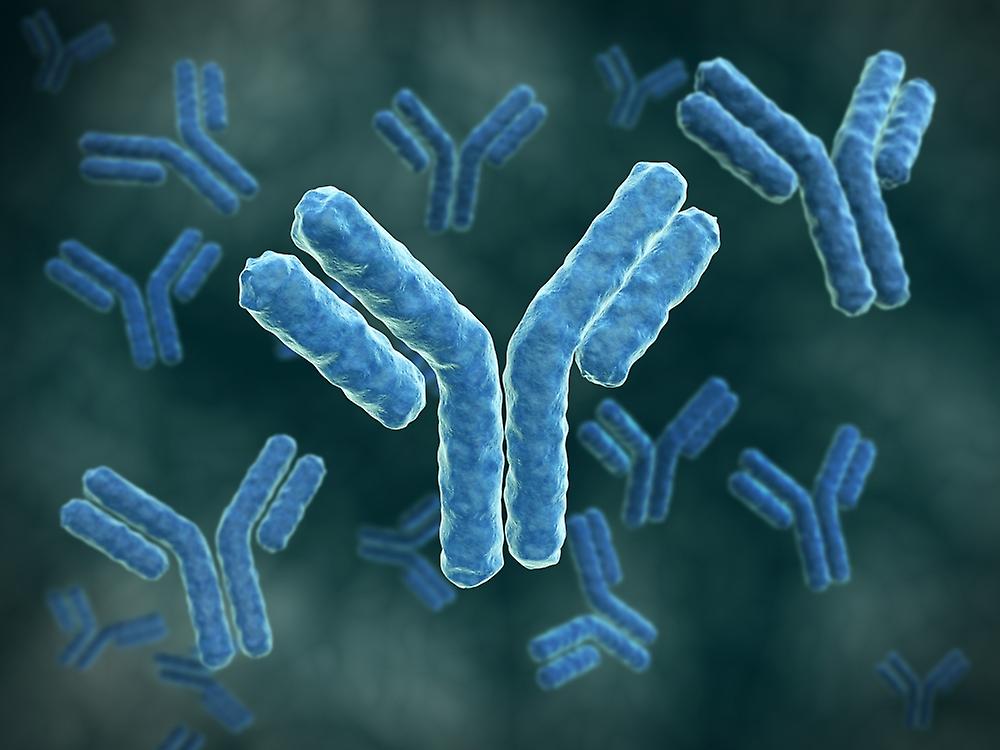
Traditionally, venomous bites from snakes, spiders, scorpions, and other creatures were met with limited treatment options. Physicians and first responders had to rely on symptomatic relief measures while hoping for the best. However, the advent of anti-venom has changed the game entirely. Anti-venom is a specially formulated serum designed to counteract the toxic effects of venom in the human body. By neutralizing the venom's components, anti-venom provides a lifeline for victims and offers them a fighting chance at survival. Its effectiveness lies in the fact that it directly targets the venom, rather than merely managing the symptoms. One of the most significant advantages of anti-venom is its rapid response time. In critical situations, every second counts, and anti-venom's quick action can mean the difference between life and death. With this breakthrough, emergency responders now have a powerful tool at their disposal to combat the devastating effects of venomous bites. Furthermore, Anti-Venom has displayed impressive efficacy across a wide range of venomous species. Whether it's a snakebite, spider bite, or scorpion sting, anti-venom has proven its versatility and effectiveness in countering the diverse toxicities present in venomous creatures. This adaptability makes anti-venom an indispensable resource in emergency rooms and healthcare facilities worldwide. The development and production of anti-venom have also seen significant advancements. Scientists and pharmaceutical companies have collaborated to create specialized formulations that target specific types of venom. This targeted approach allows for more precise and effective treatment, minimizing the risk of adverse reactions and enhancing patient outcomes. Although Anti-Venom is indeed a game-changer, it is essential to acknowledge the challenges associated with its availability and accessibility. Producing anti-venom requires complex processes, including the extraction of venom from venomous creatures, followed by purification and formulation. These processes can be time-consuming and resource-intensive, posing obstacles to widespread distribution. Moreover, anti-venom's availability varies across regions and may be scarce in certain areas, particularly those with limited healthcare infrastructure. Addressing these challenges requires a collaborative effort between governments, pharmaceutical companies, and healthcare organizations to ensure that anti-venom reaches those who need it most. Nevertheless, the impact of Anti-Venom cannot be overstated. It has transformed emergency response protocols, providing healthcare professionals with a powerful weapon to combat venomous bites effectively. Anti-venom's inclusion in medical arsenals has saved countless lives and offered hope to victims who once faced dire circumstances. As research and development continue, it is expected that Anti-Venom will further evolve, becoming even more potent and accessible. Scientists are exploring novel methods to improve the efficacy of anti-venom while streamlining its production to meet global demand. These advancements will contribute to a safer and more efficient emergency response system for venomous bites worldwide. Key pharmaceutical players are engaged in conducting clinical trials for development of Clostridium Vaccine that can offer preventive measure in the clostridium difficile infection in near future. In conclusion, the emergence of anti-venom as a revolutionary treatment for venomous bites marks a monumental turning point in emergency medicine. Its ability to neutralize venom and provide a rapid response has changed the narrative surrounding venomous encounters. With continued advancements and increased accessibility, anti-venom will continue to save lives, protecting us from the dangerous effects of venomous creatures. A big, Y- shaped protein called an Immunoglobulin (Ab), also appertained to as an Immunoglobulin (Ig), is employed by the vulnerable system to honor and destroy foreign substances including dangerous bacteria and contagions. The antigen, or distinctive patch of the infection, is recognised by the Immunoglobulin. It's possible for these two structures to bind together precisely because each tip of the" Y" of an Immunoglobulin has a paratope (like to a cinch) that's particular for one epitope (analogous to a key) on an antigen.
A pathogen or an infected cell can be marked by an Immunoglobulin for attack by other vulnerable system factors using this list process, or it can be incontinently neutralised (for illustration, by blocking a part of a contagion that's essential for its irruption). The antigen- list spots at both tips of the Immunoglobulin are inversely different, enabling the vulnerable system to honor millions of distinct antigens. The remaining portion of the Immunoglobulin, still, is frequently stable. IgA, IgD, IgE, IgG, and IgM are the only kinds that identify the Immunoglobulin's class or isotypes. Spots involved in relations with other vulnerable system rudiments can be set up in the constant region at the Immunoglobulin box. So, in addition to some structural characteristics, the class also affects the function that an Immunoglobulin initiates after attaching to an antigen. Several kinds of antibodies also differ in terms of where in the body they're released and at what stage of a vulnerable response. Antibodies are the most significant element of the adaptive vulnerable system, along with B and T cells. One form is connected to a B cell, and the other is an answerable form that’s set up in extracellular fluids like blood tube and isn't attached. The foremost form of all antibodies is connected to the face of a B cell, and they're known as B- cell receptors (BCR). When an antigen binds to a BCR, the B cell becomes activated and begins to multiply and develop into memory B cells or tube cells, which release answerable antibodies with the same paratope and allow for long- lasting impunity to the antigen. Several concealment, including blood and towel fluids, as well as answerable antibodies are produced. Antibodies are three spherical areas that roughly form a Y shape and are composed of hefty (150 kDa) proteins that are 10 nm in size. An Immunoglobulin unit in people and the maturity of mammals is made up of four polypeptide chains two identical heavy chains, two identical light chains, and two identical light chains joined by disulphide bonds. Each chain is made up of a collection of disciplines, which are all roughly 110 amino acid long and have an analogous structure. In streamlined plates, these disciplines are generally shown as blocks. Whereas heavy chains have one variable sphere VH and three to four constant disciplines CH1, CH2, light chains only have one variable sphere VH and one constant sphere CL. An Immunoglobulin is structurally divided between the Fc, which makes up the Y- shaped box, and two antigen- binding fractions (Fab), each of which contains one VL, VH, CL, and CH1 sphere. A section of the heavy chains called the hinge is located between them, and because of its inflexibility, antibodies can attach to dyads of epitopes at different distances, form complexes (similar as dimers or trimmers), and bind effector motes more readily. In a blood protein electrophoresis test, antibodies primarily resettle to the final, gamma globulin bit. Conversely, the maturity of gamma- globulins are antibodies, which is why the two terms — as well as the symbols Ig and — were formerly used interchangeably. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|



 RSS Feed
RSS Feed
