|
Golf Equipment refers to the different items used to play the sport of golf. The golf ball, golf clubs, and devices that help in the sport are examples of Golf Equipment. Golf balls were originally constructed of a hardwood, such as beech. From the 14th through the 16th century, more costly golf balls were constructed of a leather skin packed with down feathers; they were known as "featheries."
Around the mid-1800s, a new substance known as gutta-percha, which was manufactured from the latex of the East Asian sapodilla tree, began to be utilised to make more affordable golf balls which comes in Golf Equipment known as "gutties," which had comparable flight characteristics as featheries. During the game, a player often carries many Golf Equipment (but not more than fourteen, as stated by the regulations). Woods, irons, and putters are the three primary types of clubs. Woods are used for long shots from the tee or fairway, and occasionally from the rough, whereas irons are used for precise shots from the fairway and the rough. A hybrid club combines the straight-hitting capabilities of irons with the easy-hitting attributes of higher-lofted woods. For long shots from tough areas, a hybrid is frequently employed. Hybrids are also utilised by players who struggle to get the ball airborne with long irons. Wedges are irons that are used to hit shorter strokes. Wedges are used for approach shots to the green from challenging terrain such as sand or rough. Putters are usually used on the green, although they may also be handy for some approach shots. Putters have low loft, which means the ball remains near to the ground when struck. A driver, 3 and 5-woods, irons numbered 3 to 9, pitching wedge, sand wedge, and putter were once the most typical clubs in a set. When on the green, a ball may be picked up to be cleaned or if it is in the way of an opponent's putting line; there are a few additional situations in which a ball may be lifted.
0 Comments
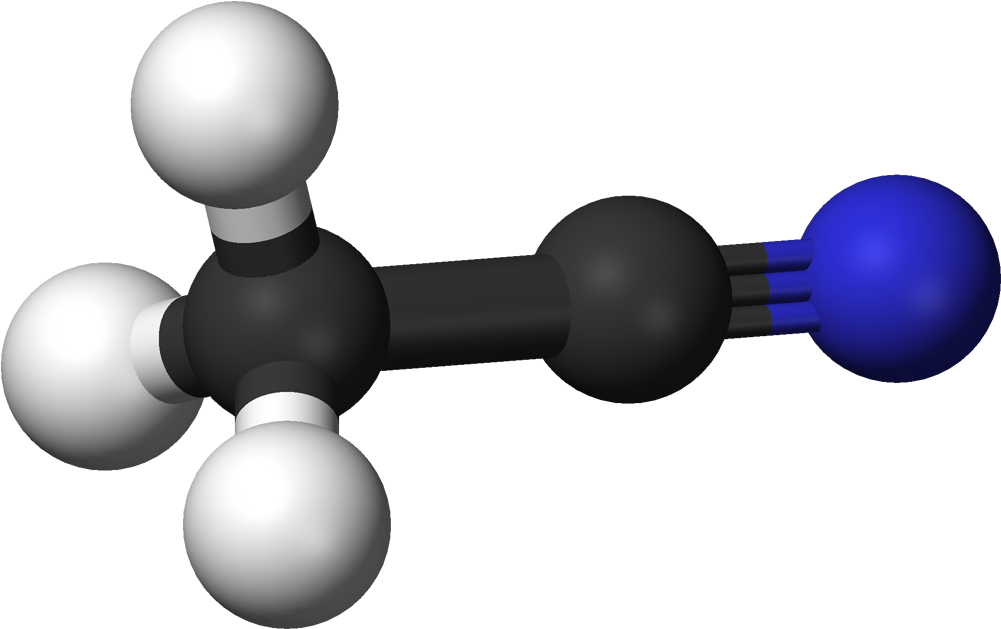
Acetonitrile Is A Nitrile Composed Of Hydrogen Cyanide With A Methyl Group In Lieu Of The Hydrogen4/24/2023 Acetonitrile functions as a polar aprotic solvent as well as an EC 3.5.1.4 (amidase) inhibitor. It is a volatile organic chemical and an aliphatic nitrile. The chemical molecule Acetonitrile, often known as MeCN (methyl cyanide), has the formula CH3CN and the structural formula H3CCN.
The simplest organic nitrile is this colourless liquid; hydrogen cyanide is another simple nitrile, but the cyanide anion is not considered to be organic. It is mostly created as a by-product of the production of acrylonitrile. It serves as a polar aprotic solvent for chemical synthesis and butadiene purification. The CN distance of the NCC skeleton is only 1.16, making it linear. Acetonitrile is primarily used as a solvent in refineries to purify butadiene. Acetonitrile is specifically injected into the top of a distillation column that contains butadiene and other hydrocarbons, and as it descends through the column, it absorbs the butadiene, which is then delivered from the bottom of the tower to a second separate tower. The butadiene is then separated using heat in the separating tower. It is employed as a medium-polarity solvent in the lab and is miscible with water and a variety of organic solvents, but not saturated hydrocarbons. Its high dielectric constant of 38.8 and convenient liquid range are both advantages. Acetonitrile, which has a dipole moment of 3.92 D, is a useful mobile phase in HPLC and LC-MS and dissolves a variety of ionic and nonpolar molecules. Due to its relatively high dielectric constant and capacity to dissolve electrolytes, it is frequently employed in battery applications. It is a common solvent in cyclic voltammetry for similar reasons. For high-performance liquid chromatography (HPLC), its UV cut-off, low viscosity, and minimal chemical reactivity make it a desirable option. As the primary solvent employed in the synthesis of oligonucleotides from nucleoside phosphoramidites, Acetonitrile plays a key role. In the manufacturing of medications and photographic film, it serves as a solvent. In the organic synthesis of several important compounds, such as acetamidine hydrochloride, thiamine, and -napthaleneacetic acid, Acetonitrile is a frequent two-carbon building block. Malononitrile is produced when it interacts with cyanogen chloride The term "food container" refers to any container, bowl, plate, tray, or other vessel used to store pre-portioned food. Although customers recognize that aesthetics and design are the most important aspects of product packaging, the truth is that health and sustainability are always present. A large variety of Food Container constructed of various materials are currently accessible in the modern developed world. Many items make use of low-density polyethylene in the form of plastic boxes. Sheet metal can be used for long-term storage or for goods that require a higher level of protection from the weather. The biscuit tin is a common example of such storage. Despite the fact that plastic Food Container are definitely convenient; nevertheless, many may contain BPA, or Biphenyl A, a chemical that inhibits and interacts with our bodies' hormones, potentially causing health problems. Manufacturers create a range of Food Container to preserve, transport, and store various food products. Food packaging often consists of a diverse range of materials that have been processed and shaped, such as plastic, metals, glass, and paper. Plastic containers, for example, are classified as stiff or flexible. Food Container may be subjected to extra preservation techniques. An automated procedure combining complicated heating and molding methods is used to create glass containers. They are microwave-safe and a common container choice since edible grade versions do not contain/transmit harmful chemicals to foods and may be reused. Manufacturers may prescribe (non-edible) desiccant packets for dry food storage to maintain freshness. Because glass is transparent and displays content, it is also often used for liquid containers. Furthermore, glass jars are frequently an appropriate choice for refrigeration and microwave cooking. Glass containers also efficiently prevent the accumulation of odors and moisture. Glass containers may be appropriate for stacking and long-term storage depending on the container form or use. Metal, particularly stainless steel, is a typical material used for bigger food processing facilities such as aseptic tanks and cubic containers, while smaller metal Food Container are also available. Metal is ideal for food storage since it is often constructed to be tamperproof in its container form. Large metal containers known as drums are commonly used in the industrial food sector to store oils and liquids. Aluminum is widely used for tray containers and is effective at preventing odor and moisture. Plastic containers are a popular alternative for airtight food storage and are frequently utilized for a variety of smaller storage needs. These containers are good for numerous applications, yet recycled plastic is not advised for food processing to minimize contamination transfer. Plastic Food Containers are useful for both wet and dry food. They are made of lightweight materials and come in rigid and semi-rigid configurations. While rigid Food Containers keep their shape and can handle a wide range of solid-formed items, semi-rigid forms are best suited for dry materials and some liquid foods. Paper containers are often used for food transportation and can hold both cold and hot items. They are also usually made to be leak resistant. They are recyclable due to their biodegradable and compostable qualities; they are typically made of cellulose paper fibers. Bulk packaging may be the most cost-effective choice for food packing. To save money and reduce trash, check into eco-friendly bulk solutions. Infant Formula (IF) is a synthetic diet that is meant to nourish new-borns up to the age of one year and can be used to partially or totally replace nursing. These are often produced for bottle-feeding and come in the form of a powder that is combined with water or another liquid and is commonly used as a replacement for breast milk.
Infant Formula makers say that the composition of their products is nearly identical to that of mother's milk. Purified whey and casein from cow's milk as a protein source, a combination of vegetable oils as a lipid source, lactose as a carbohydrate source, a mixture of vitamins and minerals, and additional elements vary depending on the producer. Although breast milk is the greatest nourishment for young children and it is not recommended to replace it with any Infant Formula, a broad selection of new-born formulas with appealing packaging are available on the market, attracting customers' interest and growing the market for Infant Formulas. Infant Formulas are specialized goods developed to meet the nutritional needs of new-born aged 0 to 12 months. To meet the various nutritional demands of new-born, specific formulas are required. Many baby health risks or fatalities might be prevented with good Infant Formula component selection. Throughout thorough processing, all of these substances must be proved to be safe for new-born feeding. Regulatory authorities cannot change the composition of authorized Infant Formula without prior approval for the processing, addition, and/or alteration of components. Infant Formulae are intended to replace or augment breast milk, and their composition should be as close to that of breast milk as feasible. To balance the fatty acid profile, the fat percentage in Infant Formula is often achieved with vegetable oil blends. As a result, the composition of Infant Formula is determined by the type and quantity of oils utilized. Another method involves changing the oil by trans-esterification and interesterification processes to alter its melting behaviour, digestion, absorption, and nutritional value to mimic breast milk fat. Infant Formula refers to goods designed for use by new-borns between the ages of 0 and 6 months, and must be acceptable for new-born from birth if they are not exclusively breast-fed. Because of the possible risks associated with nourishment for young children, the Infant Formula business is closely regulated. A variety of regulatory agencies supervise the norms and practises that corporations must follow throughout the creation, manufacturing, and commercialization of Infant Formulas. Infant Formulae are liquids or reconstituted powders that are given to new-borns and young children to replace human milk. Infant Formulae have a unique function in the diet since they are some new-borns’ only source of nutrition. Ingredients have been introduced to Infant Formula in recent decades not just to better replicate the composition of human milk, but also to provide health advantages. Fortifying recipes with iron, adding nucleotides, and modifying the composition of fat blends are some examples. Infant Formulae with extra supplies of arachidonic acid and docosahexenoic acid have recently been accessible in the United States, Europe, and other parts of the world. In the United States, novel components such as probiotics and genetically engineered chemicals are being explored for inclusion in Infant Formula. Because infancy is a very susceptible phase of fast growth and development, feeding adjustments have the potential to help or harm in the immediate term, throughout early childhood, and even into adulthood. As a result, assessments of safety parameters during infancy must be equally or even more severe than throughout other stages of the life cycle. New substances used into formulae must represent no or little harm to new-borns. Biopolymers are organic materials produced by the cells of living things. Biopolymers, like other polymers, are made up of monomeric units that are linked together by covalent bonds to create bigger molecules. Polynucleotides, polypeptides, and polysaccharides are the three primary groups of Biopolymers, which are categorized based on the monomers employed and the structure of the Biopolymers generated. Long polymers of nucleotides, such as RNA and DNA, are known as nucleotides. Proteins and shorter polymers of amino acids are examples of polypeptides; collagen, actin, and fibrin are a few well-known ones. Starch, cellulose, and alginate are a few examples of polysaccharides, which are long or branching chains of sugar carbs.
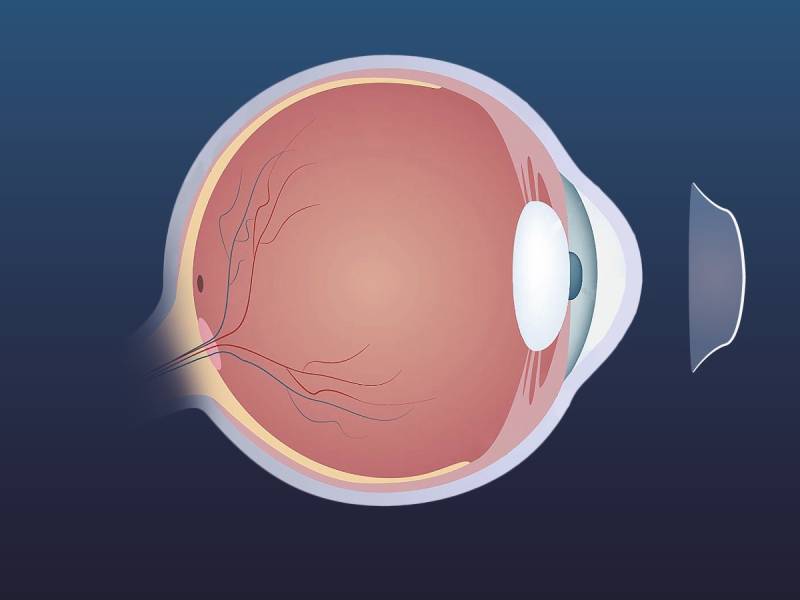
Natural rubbers, suberin and lignin (complex polyphenolic polymers), cutin and cutan (complex polymers of long-chain fatty acids), and melanin are more examples of Biopolymers. Biopolymers and synthetic polymers differ significantly from one another in terms of their structural makeup. Monomers are repeating components that make up all polymers. Although this is not a defining trait, Biopolymers frequently have a clearly defined structure (as in the case of lignocellulose): In the case of proteins, the precise chemical make-up and the order in which these units are assembled are referred to as the fundamental structure. Numerous Biopolymers spontaneously fold into recognisable compact forms that control their biological activities and intricately rely on their core structures. The study of the structural characteristics of Biopolymers is known as structural biology. Contrarily, the majority of synthetic polymers have far more straightforward and sporadic (or stochastic) architectures. Due to this feature, Biopolymers lack a molecular mass distribution. In fact, all Biopolymers of a type—let's say, one particular protein—are identical since their synthesis is governed by a template-directed mechanism in the majority of in vivo systems. They all include comparable sequences and amounts of monomers, and as a result, all of them have the same mass. In contrast to the polydispersity found in synthetic polymers, this phenomenon is known as monodispersity. Biopolymers thus have a disparity of 1.Due to their different uses in biomedicine and industry, Biopolymers have two primary application categories. Biopolymers are extensively utilised in tissue engineering, medical devices, and the pharmaceutical sector since one of the key goals of biomedical engineering is to replicate body components to maintain normal bodily functions. More precisely, because they are accessible and affordable, polypeptides like collagen and silk are being exploited in cutting-edge research as biocompatible materials. Gelatin polymer is frequently used as an adhesive while treating wounds. Gelatin-based scaffolds and films enable the scaffolds to contain medications and other nutrients that may be supplied to a wound to aid in healing. In the food business, Biopolymers are used for coating meals, edible encapsulation films, and packaging. The food sector uses polylactic acid often because of its transparent colour and water resistance. But because most polymers are hydrophilic, they begin to break down when they come into contact with moisture. Food-encapsulating edible films are another application for Biopolymers. The three Biopolymers that are most frequently used in packaging are starch, polyhydroxyalkanoate, and polylactic acid. Starch and PLA are frequently used for packaging since they are readily accessible on the market and biodegradable. Their thermal and barrier characteristics, however, are not optimal. Water can harm the contents of the package since hydrophilic polymers are not water resistant and allow water to pass through the packing. As a Biopolymers with excellent barrier properties, polyglycolic acid (PGA) is currently being employed to overcome the PLA and starch-related barrier challenges. Orthokeratology, Which Uses Specially Made Lenses Worn While The Sleeping, Enhances Your Eyesight4/19/2023 Orthokeratology, or ortho-k, is the temporary reshaping of the cornea with specifically designed and fitted contact lenses to improve eyesight. Orthokeratology is mostly used to cure myopia (near-sightedness). This eyesight impairment is generally treatable with glasses, regular contact lenses, LASIK, or PRK. Orthokeratology is a non-surgical procedure that allows some individuals to ditch their glasses and avoid wearing contact lenses all the time.
Orthokeratology is occasionally prescribed for children's eyesight correction. Some children's vision might continue to alter throughout maturity into their twenties. LASIK and other vision correction operations are not indicated until the patient's vision is steady. There is no conclusive proof that ortho-k can delay the growth of myopia in children, although it is being investigated as a potential. The cornea is a transparent, dome-shaped window at the front of the eye that focuses light onto the retina and accounts for the majority of the eye's focusing capabilities. Its tissue is very malleable. The ophthalmologist will use a corneal topographer to map and measure the surface of the cornea before designing a lens specifically for the eye. Reflecting light off the surface of the eye produces the cornea map. There is no discomfort since the machine does not come into contact with the eye. The corneal topography map reveals the form and curvature of the cornea to the ophthalmologist. The lenses function by flattening the centre of the cornea, altering the way light bends as it enters the eye. The majority of Orthokeratology lenses are worn overnight to flatten the cornea before being removed throughout the day. These are rigid, gas-permeable lenses that are strong enough to sculpt the cornea while also allowing oxygen to pass through to keep your eye healthy. When ortho-k lenses are removed, the cornea remains flattened for a short period of time, and eyesight is corrected without the use of glasses. If you stop wearing the lenses at night, your eyes will revert to their previous form, and the refractive defect will reappear. To retain the vision correction, lenses must be wear on daily basis. Although some patients see considerable vision improvement in days, it might take two weeks or longer to achieve the maximal vision correction through Orthokeratology. Orthokeratology has been linked to an increased risk of infection. This danger is especially problematic for children and teenagers, who may be less able to maintain proper hand and contact lens cleanliness than adults. In most circumstances, Orthokeratology would be a therapy of choice. There are frequently simpler, less expensive, and more well-known treatments for correcting refractive problems, including as eyeglasses. Orthokeratology, often known as ortho-k, is a non-invasive and nonsurgical method that involves the fitting of specially made contacts to a patient. The cornea is temporarily reshaped during this procedure to improve eyesight. It's sometimes likened to dental braces, which are used to modify teeth in the same way as ortho-k is used to reshape the cornea. Ortho-k is primarily used to treat myopia (near-sightedness). Wearing glasses, frequent contact lenses, laser eye surgery, or photorefractive keratectomy are all options for treating myopia. Because there is no age restriction for Orthokeratology, treatment is occasionally recommended to enhance a child's eyesight. Because some children's eyesight continues to shift until early adulthood, surgical treatments such as LASIK and PRK are not indicated for them. Furthermore, while there is no conclusive proof that ortho-k can arrest the growth of myopia in youngsters. Orthokeratology, Which Uses Specially Made Lenses Worn While The Sleeping, Enhances Your Eyesight4/19/2023 Orthokeratology, or ortho-k, is the temporary reshaping of the cornea with specifically designed and fitted contact lenses to improve eyesight. Orthokeratology is mostly used to cure myopia (near-sightedness). This eyesight impairment is generally treatable with glasses, regular contact lenses, LASIK, or PRK. Orthokeratology is a non-surgical procedure that allows some individuals to ditch their glasses and avoid wearing contact lenses all the time.
Orthokeratology is occasionally prescribed for children's eyesight correction. Some children's vision might continue to alter throughout maturity into their twenties. LASIK and other vision correction operations are not indicated until the patient's vision is steady. There is no conclusive proof that ortho-k can delay the growth of myopia in children, although it is being investigated as a potential. The cornea is a transparent, dome-shaped window at the front of the eye that focuses light onto the retina and accounts for the majority of the eye's focusing capabilities. Its tissue is very malleable. The ophthalmologist will use a corneal topographer to map and measure the surface of the cornea before designing a lens specifically for the eye. Reflecting light off the surface of the eye produces the cornea map. There is no discomfort since the machine does not come into contact with the eye. The corneal topography map reveals the form and curvature of the cornea to the ophthalmologist. The lenses function by flattening the centre of the cornea, altering the way light bends as it enters the eye. The majority of Orthokeratology lenses are worn overnight to flatten the cornea before being removed throughout the day. These are rigid, gas-permeable lenses that are strong enough to sculpt the cornea while also allowing oxygen to pass through to keep your eye healthy. When ortho-k lenses are removed, the cornea remains flattened for a short period of time, and eyesight is corrected without the use of glasses. If you stop wearing the lenses at night, your eyes will revert to their previous form, and the refractive defect will reappear. To retain the vision correction, lenses must be wear on daily basis. Although some patients see considerable vision improvement in days, it might take two weeks or longer to achieve the maximal vision correction through Orthokeratology. Orthokeratology has been linked to an increased risk of infection. This danger is especially problematic for children and teenagers, who may be less able to maintain proper hand and contact lens cleanliness than adults. In most circumstances, Orthokeratology would be a therapy of choice. There are frequently simpler, less expensive, and more well-known treatments for correcting refractive problems, including as eyeglasses. Orthokeratology, often known as ortho-k, is a non-invasive and nonsurgical method that involves the fitting of specially made contacts to a patient. The cornea is temporarily reshaped during this procedure to improve eyesight. It's sometimes likened to dental braces, which are used to modify teeth in the same way as ortho-k is used to reshape the cornea. Ortho-k is primarily used to treat myopia (near-sightedness). Wearing glasses, frequent contact lenses, laser eye surgery, or photorefractive keratectomy are all options for treating myopia. Because there is no age restriction for Orthokeratology, treatment is occasionally recommended to enhance a child's eyesight. Because some children's eyesight continues to shift until early adulthood, surgical treatments such as LASIK and PRK are not indicated for them. Furthermore, while there is no conclusive proof that ortho-k can arrest the growth of myopia in youngsters. A plant species of the holly genus Ilex endemic to South America is known as Yerba Mate or yerba-mate. Augustin Saint-Hilaire, a French botanist, gave it its name. The plant's leaves may be used to prepare the mate beverage by steeping them in hot water. It is used to produce tereré when cold brewed. Caffeine may be found in both the plant and the beverage.
Before the arrival of the Europeans to the Americas, the native Guaran and certain Tup populations were the first to plant and use Yerba Mate. Its usage was restricted to the locals of just two departments in the area that is now Paraguay, namely Amambay and Alto Paraná. Upon the Jesuits' discovery of its potential for commercialization, Yerba Mate expanded across the province and even to other parts of the Spanish Crown. Yerba Mate is historically eaten in Argentina, Uruguay, Southern Brazil, the Gran Chaco of Bolivia, and Southern Chile, as well as in the central and southern parts of South America, particularly in Paraguay. As a result of 19th-century Syrian immigration to Argentina, it has also gained popularity among the Druze and Alawite communities in the Levant, particularly in Syria and Lebanon, where it is imported from Paraguay and Argentina. IN addition to being offered as an iced tea in bottles or cans, Yerba Mate is now distributed all over the world in a variety of energy drinks. The plant is known by the Guaran name ka'a, which translates to "herb" in English. The Portuguese phrase "congonha," which refers to a number of plant species, is derived from the Tup idiom "k'gi," which roughly translates to "what keeps us alive." However, this language is no longer often used. Mate is derived from the Quechua word mati, which also means "gourd" and "container for a drink." Both contemporary Portuguese and Spanish use the word mate. The first syllable of the word "mate" is stressed. The Spanish term hierba, which is also spelt yerba in Latin America, means "herb." Yerba is a word that can mean "herb," "grass," or "weed." It could also be applied to marijuana. Yerba only refers to the Yerba Mate plant in Argentina. As a result, Yerba Mate was initially interpreted as the "gourd herb," or the herb that is consumed from cantaloupe. Lex paraguariensis starts as a shrub and develops into a tree that may reach a height of 15 metres. The evergreen leaves have serrated edges and are 7-110 millimetres long by 30-55 millimetres broad. The names yerba and erva, both of which mean "herb," are sometimes given to the leaves. They are commercially harvested, contain caffeine (also known as mateine in various regions of the world), and similar xanthine alkaloids. The blooms have four petals, are tiny, and are greenish-white. Red drupes with a diameter of 4-6 millimetres make up the fruit. The indigenous Guaran people of Paraguay's area were the first to use Yerba Mate, and it then extended to the Tup people who resided in the districts of Amambay and Alto Paraná. Its usage increased widely throughout European colonization, especially in the Spanish colony of Paraguay in the late 16th century, among both Spanish immigrants and indigenous Guaran who had already been using it to some extent before the Spanish arrival. Due to its extensive use, it became Paraguay's primary export over other goods like tobacco, and indigenous peoples' labour was utilised to harvest wild stands. In order to create Carbonated water and other carbonated beverages, the concentration of Carbonate and bi Carbonate ions in water must be increased. This can be done either by adding carbon dioxide gas under pressure or by dissolving Carbonate or bi Carbonate salts into the water. The term is also used as a verb. The term "Carbonate" in geology and mineralogy can be used to describe both Carbonate minerals and Carbonate rock (which is primarily composed of Carbonate minerals), both of which are dominated by the Carbonate ion.
In chemically precipitated sedimentary rock, Carbonate minerals are exceedingly diverse and pervasive. The Carbonate ion, a polyatomic ion with the formula CO23, is what distinguishes a Carbonate from other salts of carbonic acid. The term "Carbonate" can also refer to an organic molecule with a Carbonate group called a Carbonate ester. The simplest Oxo carbon anion is the Carbonate ion. It has D3h molecular symmetry and is made up of one carbon atom surrounded by three oxygen atoms in a trigonal planar configuration. It has a total formal charge of 2 and a molecular mass of 60.01 g/mol. It is the conjugate base of the HCO ion, the conjugate base of H2CO3, carbonic acid, which is the hydrogen Carbonate (bi Carbonate) ion. Two (long) single bonds to negative oxygen atoms make up the Carbonate ion's Lewis structure, while a single, brief double bond to a neutral oxygen atom completes it. On heating, metal Carbonates often disintegrate, releasing carbon dioxide from the long carbon cycle into the short carbon cycle and leaving a metal oxide behind. Calx, the Latin term for quicklime or calcium oxide, CaO, which is produced by roasting limestone in a lime kiln, inspired the phrase "calcination," which refers to this process. When a positively charged ion, such as M+, M2+, or M3+, develops an electrostatic attraction with the negatively charged oxygen atoms of the ion to form an ionic compound, a Carbonate salt is created. At normal pressure and temperature, the majority of Carbonate salts are insoluble in water, with solubility constants of less than 1108. Several uranium Carbonates as well as the Carbonates of lithium, sodium, potassium, rubidium, caesium, and ammonium are exceptions. Carbonate, bi Carbonate, carbon dioxide, and carbonic acid all coexist in aqueous solution in a dynamic equilibrium. The Carbonate ion predominates in very basic environments, whereas the bi Carbonate ion predominates in slightly basic settings. Aqueous carbon dioxide, CO2(aq), which is the predominant form under more acidic conditions, is in equilibrium with carbonic acid when combined with water, H2O; the balance is substantially in favour of carbon dioxide. As a result, sodium bi Carbonate is weakly basic while carbon dioxide itself is a weak acid. Sodium Carbonate is also basic. Under pressure, CO2 dissolves in water to produce Carbonated water. The equilibrium for each form of Carbonate (Carbonate, bi Carbonate, carbon dioxide, and carbonic acid) shifts when the partial pressure of CO2 is reduced, such as when a soda can is opened, until the concentration of CO2 in the solution is equal to the solubility of CO2 at that temperature and pressure. The conversion of CO2 and carbonic acid occurs more quickly in living systems thanks to the enzyme carbonic anhydrase. While the bi Carbonate salts of most metals are soluble in water, the Carbonate salts of most metals are not. The balance between Carbonate, bi Carbonate, carbon dioxide, and carbonic acid in a solution is continually shifting due to the ambient temperature and pressure. Insoluble compounds are created when metal ions interact with insoluble Carbonates, as CaCO3. This explains how hard water causes scale to accumulate inside of pipes. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|






 RSS Feed
RSS Feed
