|
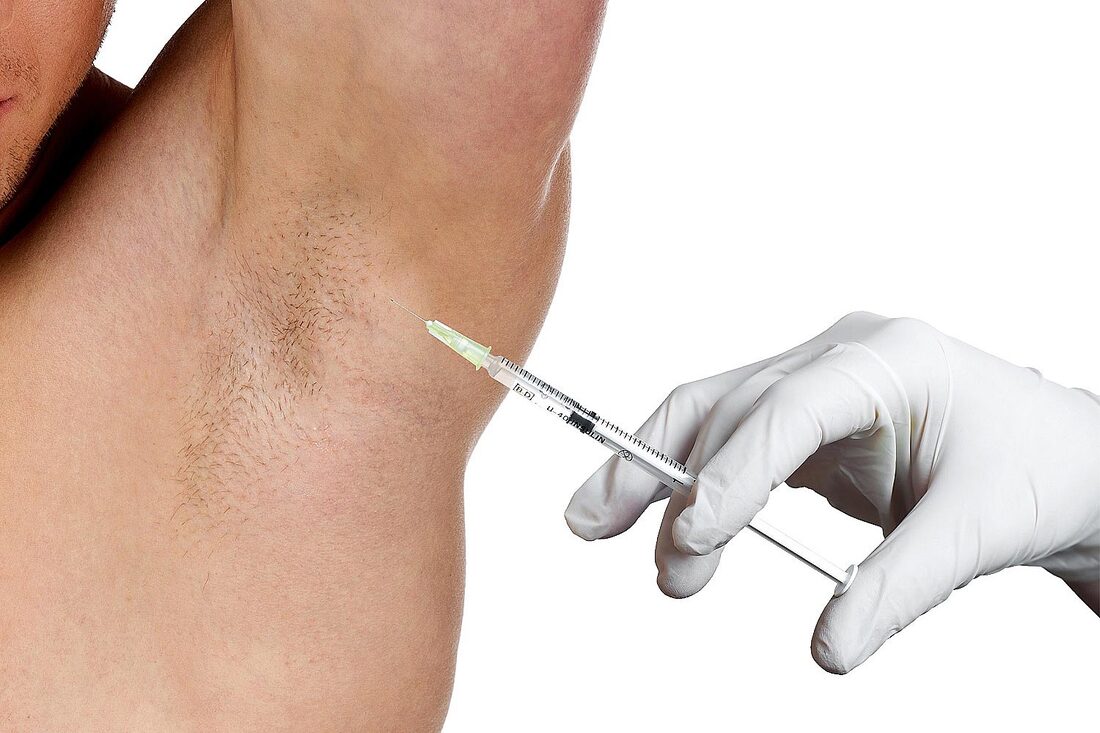
Excessive sweating can be an embarrassing and frustrating condition, impacting one's confidence and quality of life. But fear not, as advancements in hyperhidrosis treatment have made it possible to regain control over this excessive sweating and restore self-assurance. Hyperhidrosis Treatment offers a range of options tailored to individual needs, providing relief and a newfound sense of confidence. One popular approach is the use of antiperspirants specifically formulated to tackle excessive sweating. These products contain aluminum chloride, which helps to block sweat glands and reduce perspiration. Regular use of such antiperspirants can significantly decrease sweating and give you the freedom to embrace social situations without worry. For individuals seeking more long-term solutions, there are minimally invasive procedures available. One such treatment is botulinum toxin injections, commonly known as Botox. While Botox is famous for its cosmetic applications, it has also proven to be highly effective in controlling hyperhidrosis. By injecting small amounts of Botox into the affected areas, such as the underarms or palms, the nerves responsible for activating sweat glands are temporarily blocked. This results in a significant reduction in sweat production, providing much-needed relief. In more severe cases, oral medications may be prescribed as part of Hyperhidrosis Treatment. These medications work by regulating the sweat glands' activity, reducing their stimulation and subsequent sweat production. It is important to consult with a healthcare professional to determine the most suitable medication and dosage based on your specific condition and medical history. When conservative treatments do not yield the desired results, more advanced techniques like iontophoresis and laser therapy come into play. Iontophoresis involves passing a mild electric current through the affected areas, such as hands or feet, to reduce sweat gland activity. Laser therapy, on the other hand, targets and destroys sweat glands using focused laser energy. Both methods have shown promising results in managing excessive sweating and providing relief for those who have exhausted other treatment options. It's important to note that Hyperhidrosis Treatment is not a one-size-fits-all approach. Each individual's condition is unique, and a tailored treatment plan should be developed in consultation with a healthcare professional specializing in hyperhidrosis. They will assess the severity of your condition, discuss available options, and guide you towards the most suitable treatment. By exploring hyperhidrosis treatment options, individuals can reclaim their confidence, no longer held back by excessive sweating. Whether it's through antiperspirants, minimally invasive procedures, oral medications, or advanced therapies, the goal is to find the solution that works best for everyone. The growing incidences of lysosomal storage diseases among people are a major factor leading to the high demand for Lysosomal Storage Diseases Therapeutics. Remember, seeking treatment for Hyperhidrosis Treatment is not a sign of weakness or vanity but a proactive step towards improving your quality of life. So take control, regain your confidence, and embrace a sweat-free future with the help of hyperhidrosis treatment.
0 Comments
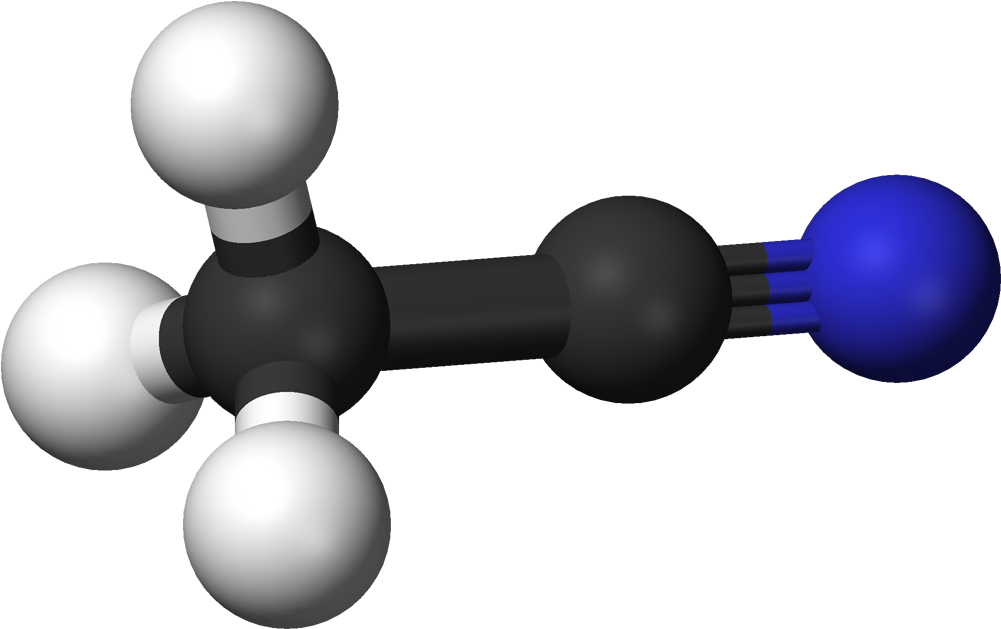
Acetonitrile Is A Nitrile Composed Of Hydrogen Cyanide With A Methyl Group In Lieu Of The Hydrogen4/24/2023 Acetonitrile functions as a polar aprotic solvent as well as an EC 3.5.1.4 (amidase) inhibitor. It is a volatile organic chemical and an aliphatic nitrile. The chemical molecule Acetonitrile, often known as MeCN (methyl cyanide), has the formula CH3CN and the structural formula H3CCN.
The simplest organic nitrile is this colourless liquid; hydrogen cyanide is another simple nitrile, but the cyanide anion is not considered to be organic. It is mostly created as a by-product of the production of acrylonitrile. It serves as a polar aprotic solvent for chemical synthesis and butadiene purification. The CN distance of the NCC skeleton is only 1.16, making it linear. Acetonitrile is primarily used as a solvent in refineries to purify butadiene. Acetonitrile is specifically injected into the top of a distillation column that contains butadiene and other hydrocarbons, and as it descends through the column, it absorbs the butadiene, which is then delivered from the bottom of the tower to a second separate tower. The butadiene is then separated using heat in the separating tower. It is employed as a medium-polarity solvent in the lab and is miscible with water and a variety of organic solvents, but not saturated hydrocarbons. Its high dielectric constant of 38.8 and convenient liquid range are both advantages. Acetonitrile, which has a dipole moment of 3.92 D, is a useful mobile phase in HPLC and LC-MS and dissolves a variety of ionic and nonpolar molecules. Due to its relatively high dielectric constant and capacity to dissolve electrolytes, it is frequently employed in battery applications. It is a common solvent in cyclic voltammetry for similar reasons. For high-performance liquid chromatography (HPLC), its UV cut-off, low viscosity, and minimal chemical reactivity make it a desirable option. As the primary solvent employed in the synthesis of oligonucleotides from nucleoside phosphoramidites, Acetonitrile plays a key role. In the manufacturing of medications and photographic film, it serves as a solvent. In the organic synthesis of several important compounds, such as acetamidine hydrochloride, thiamine, and -napthaleneacetic acid, Acetonitrile is a frequent two-carbon building block. Malononitrile is produced when it interacts with cyanogen chloride In order to create Carbonated water and other carbonated beverages, the concentration of Carbonate and bi Carbonate ions in water must be increased. This can be done either by adding carbon dioxide gas under pressure or by dissolving Carbonate or bi Carbonate salts into the water. The term is also used as a verb. The term "Carbonate" in geology and mineralogy can be used to describe both Carbonate minerals and Carbonate rock (which is primarily composed of Carbonate minerals), both of which are dominated by the Carbonate ion.
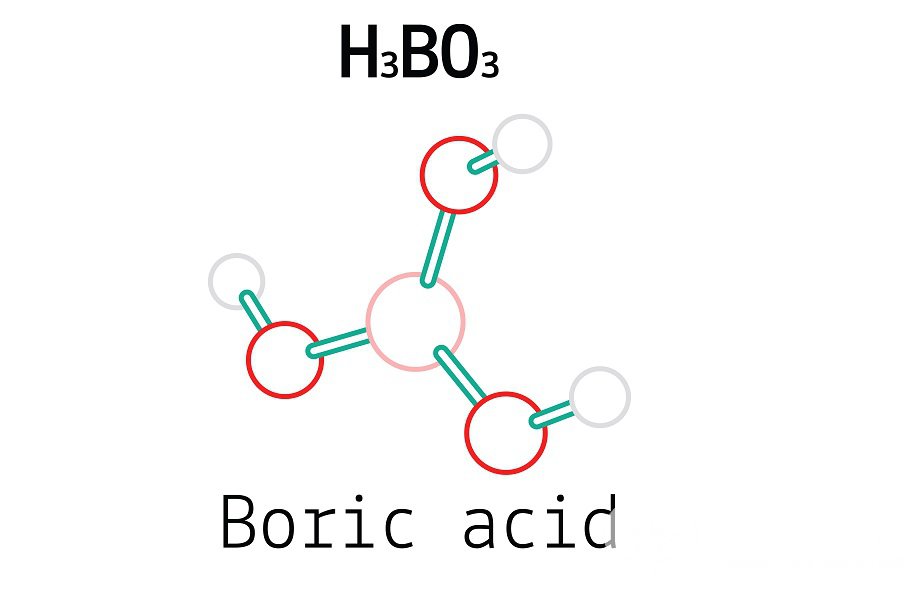
In chemically precipitated sedimentary rock, Carbonate minerals are exceedingly diverse and pervasive. The Carbonate ion, a polyatomic ion with the formula CO23, is what distinguishes a Carbonate from other salts of carbonic acid. The term "Carbonate" can also refer to an organic molecule with a Carbonate group called a Carbonate ester. The simplest Oxo carbon anion is the Carbonate ion. It has D3h molecular symmetry and is made up of one carbon atom surrounded by three oxygen atoms in a trigonal planar configuration. It has a total formal charge of 2 and a molecular mass of 60.01 g/mol. It is the conjugate base of the HCO ion, the conjugate base of H2CO3, carbonic acid, which is the hydrogen Carbonate (bi Carbonate) ion. Two (long) single bonds to negative oxygen atoms make up the Carbonate ion's Lewis structure, while a single, brief double bond to a neutral oxygen atom completes it. On heating, metal Carbonates often disintegrate, releasing carbon dioxide from the long carbon cycle into the short carbon cycle and leaving a metal oxide behind. Calx, the Latin term for quicklime or calcium oxide, CaO, which is produced by roasting limestone in a lime kiln, inspired the phrase "calcination," which refers to this process. When a positively charged ion, such as M+, M2+, or M3+, develops an electrostatic attraction with the negatively charged oxygen atoms of the ion to form an ionic compound, a Carbonate salt is created. At normal pressure and temperature, the majority of Carbonate salts are insoluble in water, with solubility constants of less than 1108. Several uranium Carbonates as well as the Carbonates of lithium, sodium, potassium, rubidium, caesium, and ammonium are exceptions. Carbonate, bi Carbonate, carbon dioxide, and carbonic acid all coexist in aqueous solution in a dynamic equilibrium. The Carbonate ion predominates in very basic environments, whereas the bi Carbonate ion predominates in slightly basic settings. Aqueous carbon dioxide, CO2(aq), which is the predominant form under more acidic conditions, is in equilibrium with carbonic acid when combined with water, H2O; the balance is substantially in favour of carbon dioxide. As a result, sodium bi Carbonate is weakly basic while carbon dioxide itself is a weak acid. Sodium Carbonate is also basic. Under pressure, CO2 dissolves in water to produce Carbonated water. The equilibrium for each form of Carbonate (Carbonate, bi Carbonate, carbon dioxide, and carbonic acid) shifts when the partial pressure of CO2 is reduced, such as when a soda can is opened, until the concentration of CO2 in the solution is equal to the solubility of CO2 at that temperature and pressure. The conversion of CO2 and carbonic acid occurs more quickly in living systems thanks to the enzyme carbonic anhydrase. While the bi Carbonate salts of most metals are soluble in water, the Carbonate salts of most metals are not. The balance between Carbonate, bi Carbonate, carbon dioxide, and carbonic acid in a solution is continually shifting due to the ambient temperature and pressure. Insoluble compounds are created when metal ions interact with insoluble Carbonates, as CaCO3. This explains how hard water causes scale to accumulate inside of pipes. Boric Acid is a weak boron acid that’s sometimes employed as an antiseptic, fungicide, honey retardant, neutron absorber, and as a precursor to other chemical composites. It's also constantly appertained to as hydrogen borate, boracic acid, ortho Boric Acid, or acid boricum. Boron, oxygen, and hydrogen combine to induce the chemical ortho Boric Acid, which has the formula B (OH) 3. Other names for it include boracic acid, trihydroxidoboron, and hydrogen orthoborate.
It occurs naturally as the mineral sassolite and is frequently set up as colourless chargers or a white greasepaint that dissolves in water. It's a weak acid that may reply with alcohols to produce borate esters as well as a variety of borate anions and mariners. As an antiseptic, fungicide, honey retardant, neutron absorber, and precursor to colourful boron composites, Boric Acid is constantly employed. Any boron oxoacid, including tetra Boric Acid H2B4O7 and Meta Boric Acid HBO2, is appertained to as" Boric Acid" in this sense. By using mineral acids, Wilhelm Homberg( 1652 – 1715) created ortho Boric Acid for the first time from borax, earning the designation Sal sedativum Hombergi(" dreamy swab of Homberg"). Nonetheless, drawing, food preservation, and other uses for borates and Boric Acid date back to the time of the ancient Greeks. Boric Acid is only dangerous if ingested internally or breathed in significant quantities, according to the mammalian standard murderous cure (LD50) estimate of, 660 mg/ kg body mass. According to the 14th edition of the Merck Index, oral boluses of Boric Acid to rats have an LD50 of5.14 g/ kg, and boluses of 5 to 20 g/ kg have been known to beget adult mortal losses. At the lower 5g limit, 350g might affect in mortal mortality for a 70 kg grown-up. Boric Acid sometimes appears in dressings and dressings like boracic fur and can be used as an antiseptic for small be or injuries. As an eye marshland, Boric Acid is used in an important adulterated result. “Boric Acid exposure that lasts a long time can damage the feathers and eventually lead to renal failure. Studies on tykes revealed testicular atrophy following exposure to 32 mg/ kg BW/ day for 90 days, despite the fact that it doesn't appear to be carcinogenic. The list of chemical complements used for hydraulic fracturing (fracking) in the Marcellus Shale in Pennsylvania also includes Boric Acid. To manage the density and rheology of the fracking fluid fitted at high pressure in the well, it’s constantly employed in confluence with guar goo as across-linking and gelatinizing agent. In fact, it’s pivotal to regulate the fluid’s density to keep the propping agent grains suspended across lengthy transport lengths so they can keep the shales’ crevices sufficiently open to allow for the birth of gas when the hydraulic pressure is released. The pH position has a significant impact on the rheological characteristics of borate cross-linked guar goo hydrogel. Certain expatriation- type electrical fuses employ Boric Acid as a de- ionization/ extinguishing agent. The decomposition and quick spring- loaded separation of the fusible element, which is typically a specific essence rod that passes through a compressed mass of Boric Acid within the fuse assembly, during an electrical fault in an expatriation- type fuse generates a tube bow. Boric Acid snappily breaks down into water vapour and boric anhydride in the high- temperature tube. As a result, the tube isde-ionized, which aids in interposing the electrical fault. Both candidiasis brought on by non-albicans candida and bacterial vaginosis brought on by excessive alkalinity can be treated with diluted Boric Acid administered as a vaginal douche. Boric Acid significantly spares vaginal lactobacilli. It is being developed as TOL-463, an intravaginal drug to treat bacterial vaginosis and vulvovaginal candidiasis. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|

 RSS Feed
RSS Feed
