|
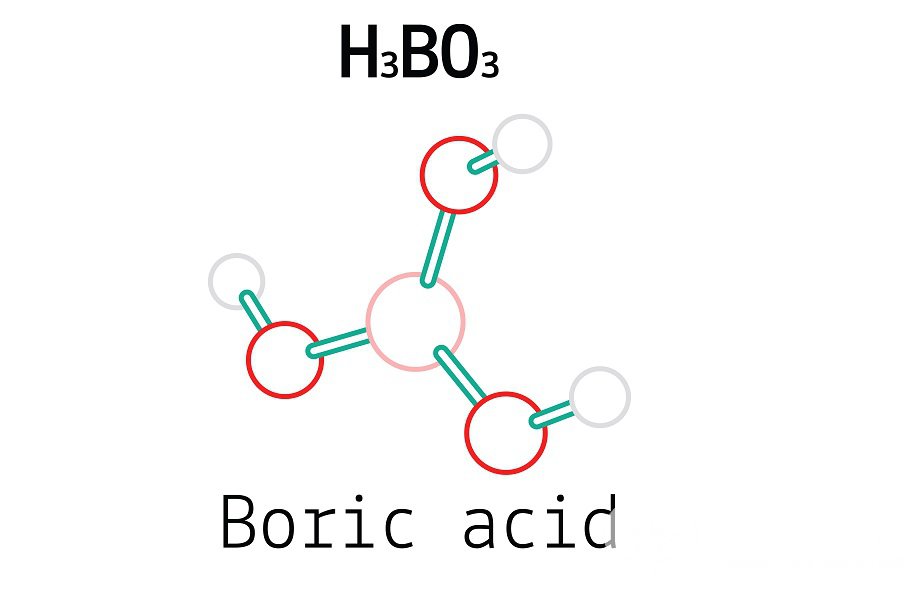
Boric Acid is a weak boron acid that’s sometimes employed as an antiseptic, fungicide, honey retardant, neutron absorber, and as a precursor to other chemical composites. It's also constantly appertained to as hydrogen borate, boracic acid, ortho Boric Acid, or acid boricum. Boron, oxygen, and hydrogen combine to induce the chemical ortho Boric Acid, which has the formula B (OH) 3. Other names for it include boracic acid, trihydroxidoboron, and hydrogen orthoborate.
It occurs naturally as the mineral sassolite and is frequently set up as colourless chargers or a white greasepaint that dissolves in water. It's a weak acid that may reply with alcohols to produce borate esters as well as a variety of borate anions and mariners. As an antiseptic, fungicide, honey retardant, neutron absorber, and precursor to colourful boron composites, Boric Acid is constantly employed. Any boron oxoacid, including tetra Boric Acid H2B4O7 and Meta Boric Acid HBO2, is appertained to as" Boric Acid" in this sense. By using mineral acids, Wilhelm Homberg( 1652 – 1715) created ortho Boric Acid for the first time from borax, earning the designation Sal sedativum Hombergi(" dreamy swab of Homberg"). Nonetheless, drawing, food preservation, and other uses for borates and Boric Acid date back to the time of the ancient Greeks. Boric Acid is only dangerous if ingested internally or breathed in significant quantities, according to the mammalian standard murderous cure (LD50) estimate of, 660 mg/ kg body mass. According to the 14th edition of the Merck Index, oral boluses of Boric Acid to rats have an LD50 of5.14 g/ kg, and boluses of 5 to 20 g/ kg have been known to beget adult mortal losses. At the lower 5g limit, 350g might affect in mortal mortality for a 70 kg grown-up. Boric Acid sometimes appears in dressings and dressings like boracic fur and can be used as an antiseptic for small be or injuries. As an eye marshland, Boric Acid is used in an important adulterated result. “Boric Acid exposure that lasts a long time can damage the feathers and eventually lead to renal failure. Studies on tykes revealed testicular atrophy following exposure to 32 mg/ kg BW/ day for 90 days, despite the fact that it doesn't appear to be carcinogenic. The list of chemical complements used for hydraulic fracturing (fracking) in the Marcellus Shale in Pennsylvania also includes Boric Acid. To manage the density and rheology of the fracking fluid fitted at high pressure in the well, it’s constantly employed in confluence with guar goo as across-linking and gelatinizing agent. In fact, it’s pivotal to regulate the fluid’s density to keep the propping agent grains suspended across lengthy transport lengths so they can keep the shales’ crevices sufficiently open to allow for the birth of gas when the hydraulic pressure is released. The pH position has a significant impact on the rheological characteristics of borate cross-linked guar goo hydrogel. Certain expatriation- type electrical fuses employ Boric Acid as a de- ionization/ extinguishing agent. The decomposition and quick spring- loaded separation of the fusible element, which is typically a specific essence rod that passes through a compressed mass of Boric Acid within the fuse assembly, during an electrical fault in an expatriation- type fuse generates a tube bow. Boric Acid snappily breaks down into water vapour and boric anhydride in the high- temperature tube. As a result, the tube isde-ionized, which aids in interposing the electrical fault. Both candidiasis brought on by non-albicans candida and bacterial vaginosis brought on by excessive alkalinity can be treated with diluted Boric Acid administered as a vaginal douche. Boric Acid significantly spares vaginal lactobacilli. It is being developed as TOL-463, an intravaginal drug to treat bacterial vaginosis and vulvovaginal candidiasis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|
 RSS Feed
RSS Feed
