|
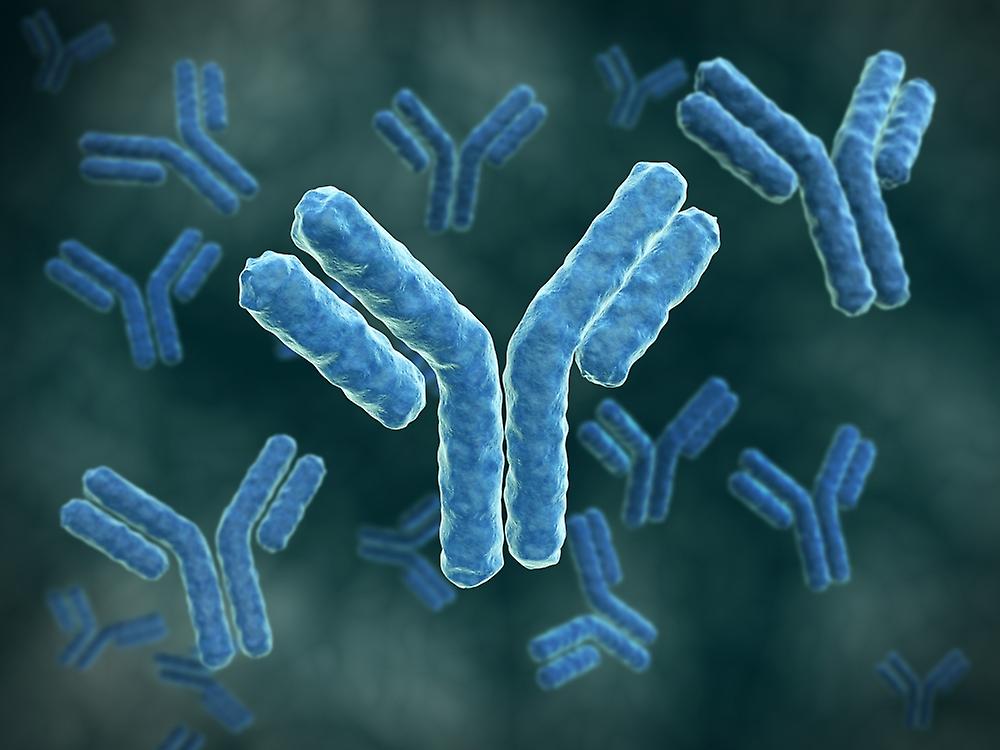
A big, Y- shaped protein called an Immunoglobulin (Ab), also appertained to as an Immunoglobulin (Ig), is employed by the vulnerable system to honor and destroy foreign substances including dangerous bacteria and contagions. The antigen, or distinctive patch of the infection, is recognised by the Immunoglobulin. It's possible for these two structures to bind together precisely because each tip of the" Y" of an Immunoglobulin has a paratope (like to a cinch) that's particular for one epitope (analogous to a key) on an antigen.
A pathogen or an infected cell can be marked by an Immunoglobulin for attack by other vulnerable system factors using this list process, or it can be incontinently neutralised (for illustration, by blocking a part of a contagion that's essential for its irruption). The antigen- list spots at both tips of the Immunoglobulin are inversely different, enabling the vulnerable system to honor millions of distinct antigens. The remaining portion of the Immunoglobulin, still, is frequently stable. IgA, IgD, IgE, IgG, and IgM are the only kinds that identify the Immunoglobulin's class or isotypes. Spots involved in relations with other vulnerable system rudiments can be set up in the constant region at the Immunoglobulin box. So, in addition to some structural characteristics, the class also affects the function that an Immunoglobulin initiates after attaching to an antigen. Several kinds of antibodies also differ in terms of where in the body they're released and at what stage of a vulnerable response. Antibodies are the most significant element of the adaptive vulnerable system, along with B and T cells. One form is connected to a B cell, and the other is an answerable form that’s set up in extracellular fluids like blood tube and isn't attached. The foremost form of all antibodies is connected to the face of a B cell, and they're known as B- cell receptors (BCR). When an antigen binds to a BCR, the B cell becomes activated and begins to multiply and develop into memory B cells or tube cells, which release answerable antibodies with the same paratope and allow for long- lasting impunity to the antigen. Several concealment, including blood and towel fluids, as well as answerable antibodies are produced. Antibodies are three spherical areas that roughly form a Y shape and are composed of hefty (150 kDa) proteins that are 10 nm in size. An Immunoglobulin unit in people and the maturity of mammals is made up of four polypeptide chains two identical heavy chains, two identical light chains, and two identical light chains joined by disulphide bonds. Each chain is made up of a collection of disciplines, which are all roughly 110 amino acid long and have an analogous structure. In streamlined plates, these disciplines are generally shown as blocks. Whereas heavy chains have one variable sphere VH and three to four constant disciplines CH1, CH2, light chains only have one variable sphere VH and one constant sphere CL. An Immunoglobulin is structurally divided between the Fc, which makes up the Y- shaped box, and two antigen- binding fractions (Fab), each of which contains one VL, VH, CL, and CH1 sphere. A section of the heavy chains called the hinge is located between them, and because of its inflexibility, antibodies can attach to dyads of epitopes at different distances, form complexes (similar as dimers or trimmers), and bind effector motes more readily. In a blood protein electrophoresis test, antibodies primarily resettle to the final, gamma globulin bit. Conversely, the maturity of gamma- globulins are antibodies, which is why the two terms — as well as the symbols Ig and — were formerly used interchangeably.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|
 RSS Feed
RSS Feed
